KL2.1 in complex with CRM1-Ran-RanBP1
Sun, Q., Jian, L.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| GTP-binding nuclear protein Ran | 216 | Homo sapiens | Mutation(s): 2 Gene Names: RAN, ARA24, OK/SW-cl.81 EC: 3.6.5 | 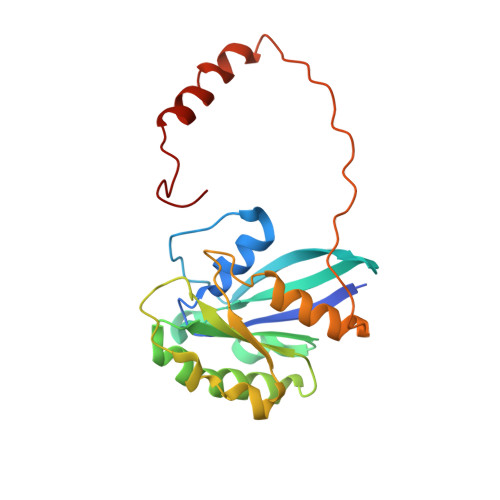 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P62826 GTEx: ENSG00000132341 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P62826 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| YRB1 isoform 1 | 140 | Saccharomyces cerevisiae | Mutation(s): 0 Gene Names: YRB1, GI526_G0000915 | 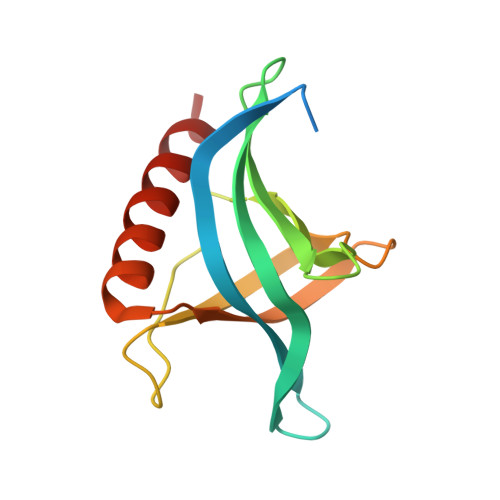 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P41920 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| CRM1 isoform 1 | 1,003 | Saccharomyces cerevisiae | Mutation(s): 11 Gene Names: CRM1, GI526_G0002640, PACBIOSEQ_LOCUS2878, PACBIOSEQ_LOCUS3002 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P30822 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 8 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GTP Download:Ideal Coordinates CCD File | K [auth A] | GUANOSINE-5'-TRIPHOSPHATE C10 H16 N5 O14 P3 XKMLYUALXHKNFT-UUOKFMHZSA-N |  | ||
| Q73 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | IA [auth C] | (3~{S},5~{R},6~{E},8~{Z},10~{R},12~{E},14~{E},16~{S})-3,16-bis(azanyl)-8,10,12-trimethyl-16-[(2~{S},4~{R},5~{S},6~{S})-5-methyl-4-oxidanyl-6-[(~{E})-prop-1-enyl]oxan-2-yl]-5-oxidanyl-hexadeca-6,8,12,14-tetraenoic acid C28 H46 N2 O5 ZMTRMZNTBHTQBT-XQYVQVJZSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | H [auth A], O [auth C], Q [auth C], Y [auth C], Z [auth C] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| DMS Download:Ideal Coordinates CCD File | AA [auth C] CA [auth C] F [auth A] FA [auth C] HA [auth C] | DIMETHYL SULFOXIDE C2 H6 O S IAZDPXIOMUYVGZ-UHFFFAOYSA-N |  | ||
| NO3 Download:Ideal Coordinates CCD File | BA [auth C] DA [auth C] E [auth A] EA [auth C] G [auth A] | NITRATE ION N O3 NHNBFGGVMKEFGY-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | GA [auth C] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| CL Download:Ideal Coordinates CCD File | M [auth C] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| MG Download:Ideal Coordinates CCD File | D [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 106.014 | α = 90 |
| b = 106.014 | β = 90 |
| c = 305.893 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data scaling |
| HKL-2000 | data reduction |
| MOLREP | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Not funded | -- |