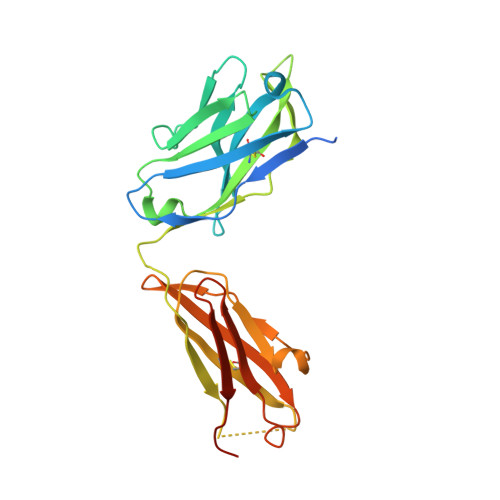
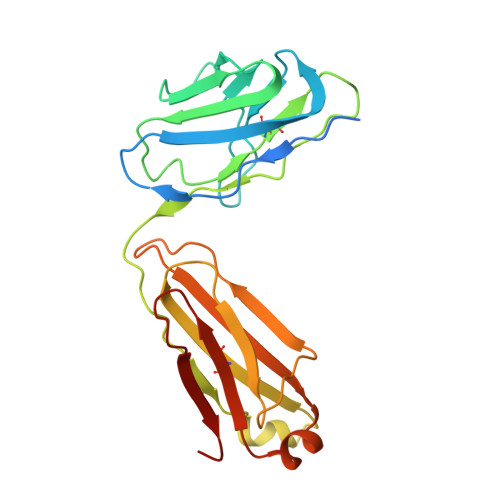
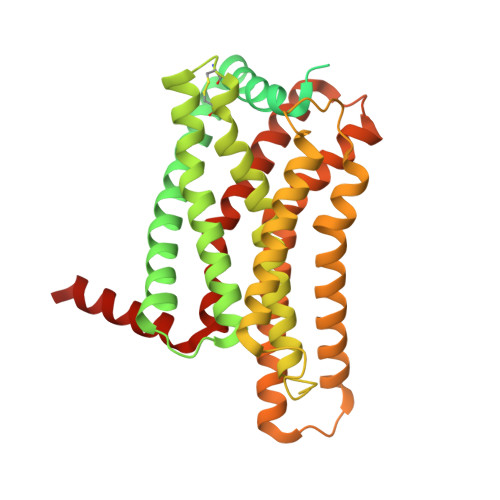
The activation mechanism and antibody binding mode for orphan GPR20.
Lin, X., Jiang, S., Wu, Y., Wei, X., Han, G.W., Wu, L., Liu, J., Chen, B., Zhang, Z., Zhao, S., Cherezov, V., Xu, F.(2023) Cell Discov 9: 23-23
- PubMed: 36849514 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41421-023-00520-8
- Primary Citation Related Structures:
8HS2, 8HS3, 8HSC - PubMed Abstract:
GPR20 is a class-A orphan G protein-coupled receptor (GPCR) and a potential therapeutic target for gastrointestinal stromal tumors (GIST) owing to its differentially high expression. An antibody-drug conjugate (ADC) containing a GPR20-binding antibody (Ab046) was recently developed in clinical trials for GIST treatment. GPR20 constitutively activates Gi proteins in the absence of any known ligand, but it remains obscure how this high basal activity is achieved. Here we report three cryo-EM structures of human GPR20 complexes including Gi-coupled GPR20 in the absence or presence of the Fab fragment of Ab046 and Gi-free GPR20. Remarkably, the structures demonstrate a uniquely folded N-terminal helix capping onto the transmembrane domain and our mutagenesis study suggests a key role of this cap region in stimulating the basal activity of GPR20. We also uncover the molecular interactions between GPR20 and Ab046, which may enable the design of tool antibodies with enhanced affinity or new functionality for GPR20. Furthermore, we report the orthosteric pocket occupied by an unassigned density which might be essential for exploring opportunities for deorphanization.
- iHuman Institute, ShanghaiTech University, Pudong, Shanghai, China.
Organizational Affiliation: