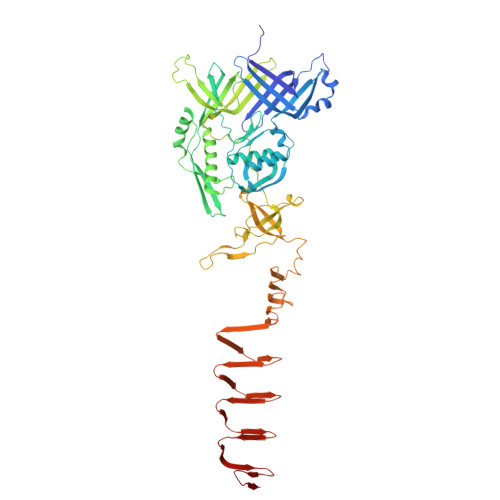
Structure and assembly of type VI secretion system cargo delivery vehicle.
He, W., Wu, K., Ouyang, Z., Bai, Y., Luo, W., Wu, D., An, H., Guo, Y., Jiao, M., Qin, Q., Zhang, J., Wu, Y., She, J., Hwang, P.M., Zheng, F., Zhu, L., Wen, Y.(2023) Cell Rep 42: 112781-112781
- PubMed: 37421630 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2023.112781
- Primary Citation Related Structures:
7YW0, 8GRA - PubMed Abstract:
Type VI secretion system is widely used in Gram-negative bacteria for injecting toxic effectors into neighboring prokaryotic or eukaryotic cells. Various effectors can be loaded onto the T6SS delivery tube via its core components: Hcp, VgrG, or PAAR. Here, we report 2.8-Å resolution cryo-EM structure of intact T6SS Hcp5-VgrG-PAAR cargo delivery system and crystal structure of unbound Hcp5 from B. fragilis NCTC 9343. Loading of Hcp5 hexameric ring onto VgrG causes expansion of its inner cavity and external surface, explaining how structural changes could be propagated to regulate co-polymerization and surrounding contractile sheath. High-affinity binding between Hcp and VgrG causes entropically unfavorable structuring of long loops. Furthermore, interactions between VgrG trimer and Hcp hexamer are asymmetric, with three of the six Hcp monomers exhibiting a major loop flip. Our study provides insights into the assembly, loading, and firing of T6SS nanomachine that contributes to bacterial inter-species competition and host interactions.
- Center for Microbiome Research of Med-X Institute, Shaanxi Provincial Key Laboratory of Sepsis in Critical Care Medicine, The First Affiliated Hospital, Xi'an Jiaotong University, Xi'an 710061, China.
Organizational Affiliation: