Novel cofactor binding motifs, electron transfer and ion pumping mechanisms of the respiratory complex NQR
Juarez, O., Fuller, J.R.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit A | 446 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrA, D6U24_04460, EYB64_17955, FLM02_04825 EC: 7.2.1.1 | 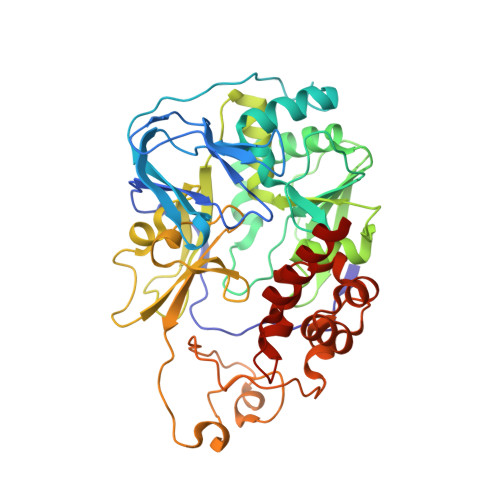 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5X1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit B | 415 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrB, D6U24_04465, ERS013186_02082, ERS013198_02508, ERS013199_02395, ERS013200_04117, ERS013202_01883, ERS013206_02987, ERS013207_01958, EYB64_17950... EC: 7.2.1.1 Membrane Entity: Yes | 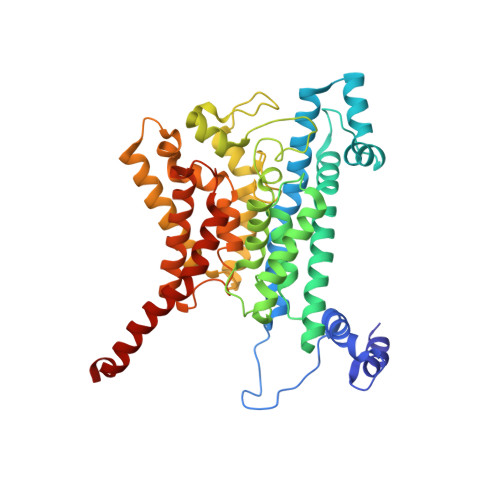 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5X0 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit C | 257 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrC, BC353_01370, D6U24_04470, ERS013165_00616, ERS013186_02083, ERS013198_02507, ERS013199_02396, ERS013200_04118, ERS013201_01110, ERS013202_01884... EC: 7.2.1.1 Membrane Entity: Yes | 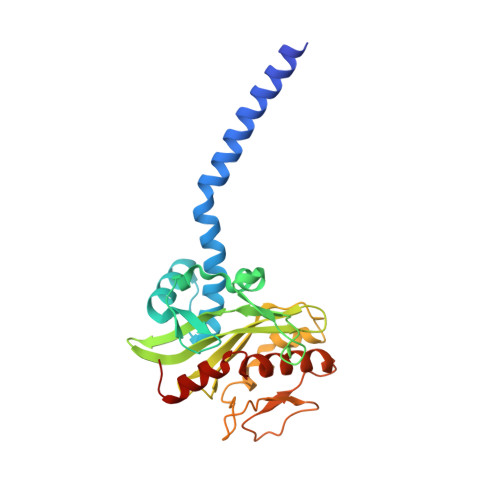 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5Y7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit D | 210 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrD, VC0395_A1881, VC395_2408 EC: 7.2.1.1 Membrane Entity: Yes | 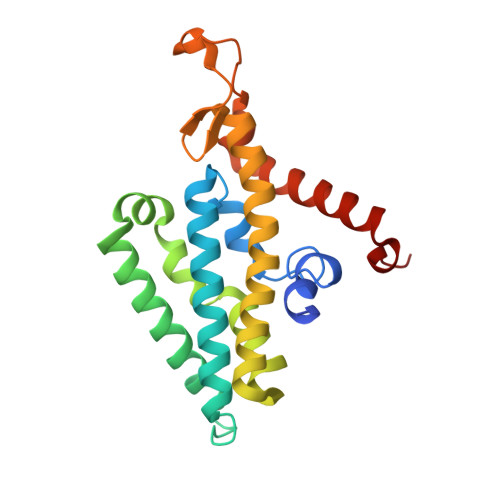 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5Y6 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 5 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit E | 198 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrE, BC353_01360, D6U24_04480, ERS013165_00614, ERS013186_02085, ERS013198_02505, ERS013199_02398, ERS013200_04120, ERS013201_01112, ERS013202_01886... EC: 7.2.1.1 Membrane Entity: Yes |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5Y5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 6 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Na(+)-translocating NADH-quinone reductase subunit F | 408 | Vibrio cholerae O395 | Mutation(s): 0 Gene Names: nqrF, D6U24_04485, ERS013198_02504, ERS013199_02399, ERS013201_01113, ERS013202_01887, ERS013206_02991, EYB64_17930, FLM12_12900 EC: 7.2.1.1 Membrane Entity: Yes |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A5F5Y4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FMN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth B], J [auth C] | FLAVIN MONONUCLEOTIDE C17 H21 N4 O9 P FVTCRASFADXXNN-SCRDCRAPSA-N |  | ||
| RBF (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | H [auth B] | RIBOFLAVIN C17 H20 N4 O6 AUNGANRZJHBGPY-SCRDCRAPSA-N |  | ||
| UQ1 (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | I [auth B] | UBIQUINONE-1 C14 H18 O4 SOECUQMRSRVZQQ-UHFFFAOYSA-N |  | ||
| FES (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | K [auth E] | FE2/S2 (INORGANIC) CLUSTER Fe2 S2 NIXDOXVAJZFRNF-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | RELION | 4.0beta2 |
| MODEL REFINEMENT | PHENIX | |
| MODEL REFINEMENT | ISOLDE | |
| MODEL REFINEMENT | Rosetta |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) | United States | AI151152 |