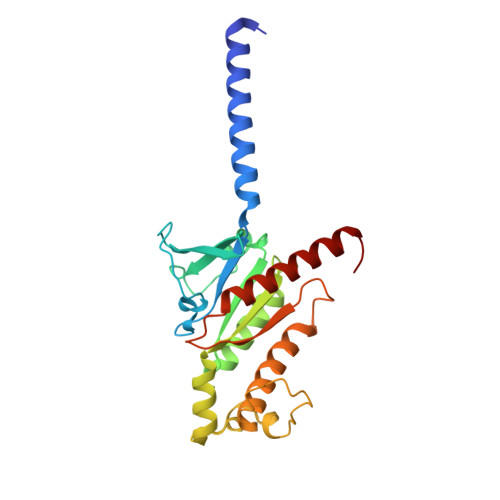
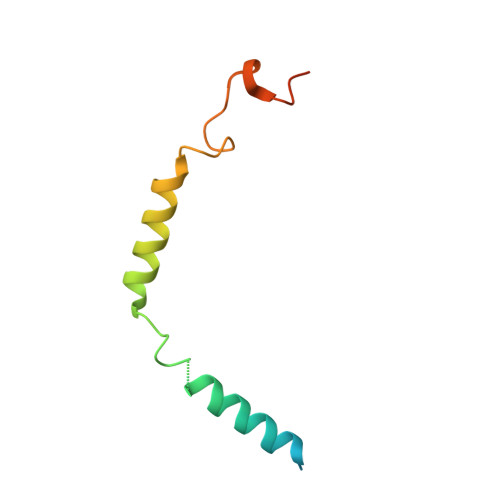
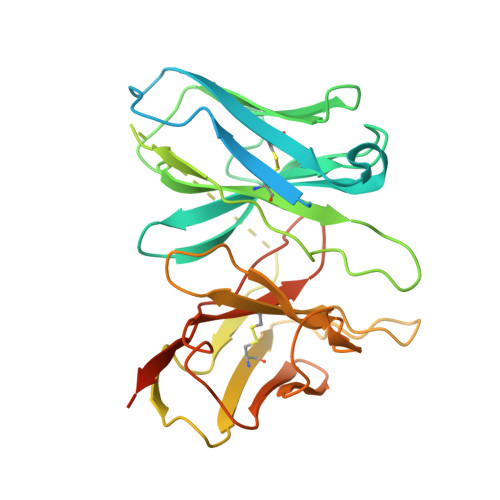
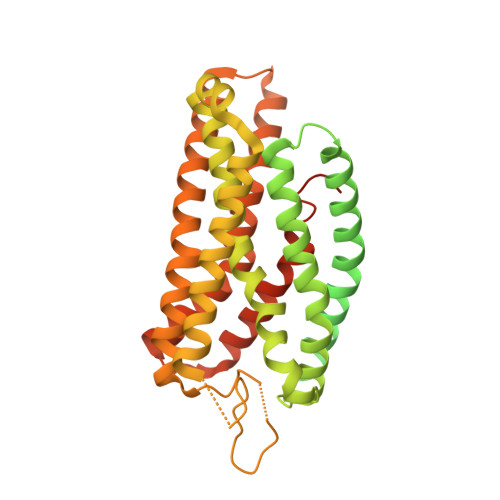
Molecular mechanism of fatty acid activation of FFAR1.
Kumari, P., Inoue, A., Chapman, K., Lian, P., Rosenbaum, D.M.(2023) Proc Natl Acad Sci U S A 120: e2219569120-e2219569120
- PubMed: 37216523 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2219569120
- Primary Citation Related Structures:
8EIT, 8EJC, 8EJK - PubMed Abstract:
FFAR1 is a G-protein-coupled receptor (GPCR) that responds to circulating free fatty acids to enhance glucose-stimulated insulin secretion and release of incretin hormones. Due to the glucose-lowering effect of FFAR1 activation, potent agonists for this receptor have been developed for the treatment of diabetes. Previous structural and biochemical studies of FFAR1 showed multiple sites of ligand binding to the inactive state but left the mechanism of fatty acid interaction and receptor activation unknown. We used cryo-electron microscopy to elucidate structures of activated FFAR1 bound to a G q mimetic, which were induced either by the endogenous FFA ligand docosahexaenoic acid or γ-linolenic acid and the agonist drug TAK-875. Our data identify the orthosteric pocket for fatty acids and show how both endogenous hormones and synthetic agonists induce changes in helical packing along the outside of the receptor that propagate to exposure of the G-protein-coupling site. These structures show how FFAR1 functions without the highly conserved "DRY" and "NPXXY" motifs of class A GPCRs and also illustrate how the orthosteric site of a receptor can be bypassed by membrane-embedded drugs to confer full activation of G protein signaling.
- Department of Biophysics, The University of Texas Southwestern Medical Center, Dallas, TX 75390.
Organizational Affiliation: