Crystal structure of putative nitroreductase from Salmonella enterica
Chang, C., Skarina, T., Mesa, N., Joachimiak, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Putative NAD(P)H nitroreductase | 183 | Salmonella enterica subsp. enterica serovar Typhimurium str. SL1344 | Mutation(s): 0 Gene Names: ydjA, SL1344_1231, G1W50_15780, G1W53_02785, G1W54_02785, G1W56_06140, G1W59_21080, G1W62_10980, G1W63_02785, G1W64_08545... EC: 1 | 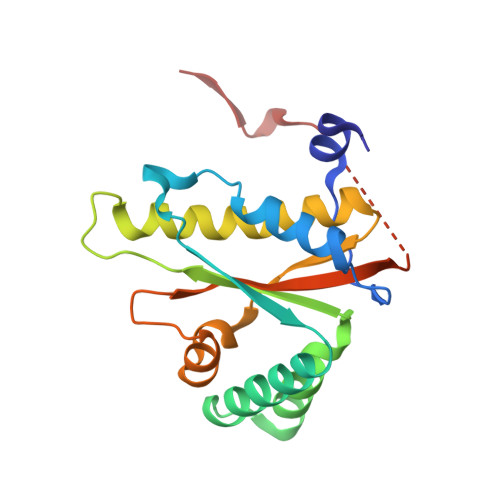 | |
UniProt | |||||
Find proteins for A0A0H3NAL3 (Salmonella typhimurium (strain SL1344)) Explore A0A0H3NAL3 Go to UniProtKB: A0A0H3NAL3 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0H3NAL3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FMN Download:Ideal Coordinates CCD File | CA [auth E] G [auth A] IA [auth F] L [auth B] Q [auth C] | FLAVIN MONONUCLEOTIDE C17 H21 N4 O9 P FVTCRASFADXXNN-SCRDCRAPSA-N |  | ||
| CIT Download:Ideal Coordinates CCD File | DA [auth E] H [auth A] JA [auth F] M [auth B] R [auth C] | CITRIC ACID C6 H8 O7 KRKNYBCHXYNGOX-UHFFFAOYSA-N |  | ||
| PGE Download:Ideal Coordinates CCD File | EA [auth E], I [auth A], MA [auth F], U [auth C], Z [auth D] | TRIETHYLENE GLYCOL C6 H14 O4 ZIBGPFATKBEMQZ-UHFFFAOYSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | AA [auth D] FA [auth E] HA [auth E] K [auth A] KA [auth F] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | BA [auth D] GA [auth E] J [auth A] N [auth B] O [auth B] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 42.588 | α = 90 |
| b = 72.719 | β = 93.94 |
| c = 172.854 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SBC-Collect | data collection |
| PHENIX | refinement |
| HKL-3000 | data scaling |
| PDB_EXTRACT | data extraction |
| HKL-3000 | data reduction |
| HKL-3000 | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Not funded | -- |