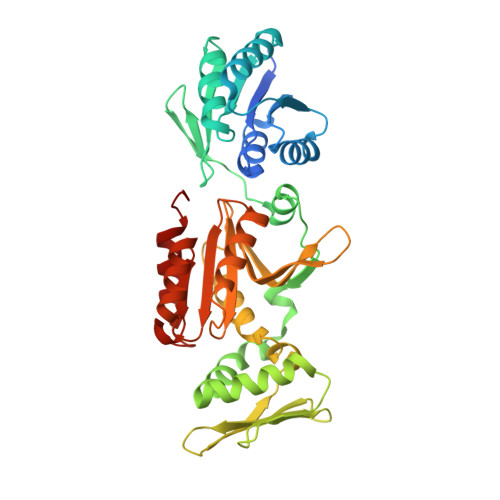
Reverse transcriptases prime DNA synthesis.
Zabrady, M., Zabrady, K., Li, A.W.H., Doherty, A.J.(2023) Nucleic Acids Res 51: 7125-7142
- PubMed: 37279911 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkad478
- Primary Citation Related Structures:
8BAO, 8BGJ - PubMed Abstract:
The discovery of reverse transcriptases (RTs) challenged the central dogma by establishing that genetic information can also flow from RNA to DNA. Although they act as DNA polymerases, RTs are distantly related to replicases that also possess de novo primase activity. Here we identify that CRISPR associated RTs (CARTs) directly prime DNA synthesis on both RNA and DNA. We demonstrate that RT-dependent priming is utilized by some CRISPR-Cas complexes to synthesise new spacers and integrate these into CRISPR arrays. Expanding our analyses, we show that primer synthesis activity is conserved in representatives of other major RT classes, including group II intron RT, telomerase and retroviruses. Together, these findings establish a conserved innate ability of RTs to catalyse de novo DNA primer synthesis, independently of accessory domains or alternative priming mechanisms, which likely plays important roles in a wide variety of biological pathways.
- Genome Damage and Stability Centre, School of Life Sciences, University of Sussex, Brighton BN1 9RQ, UK.
Organizational Affiliation: