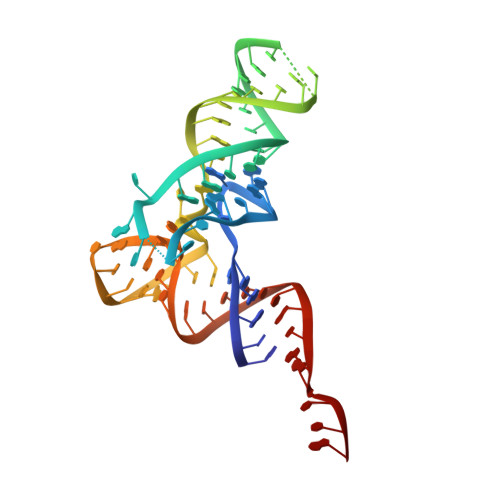
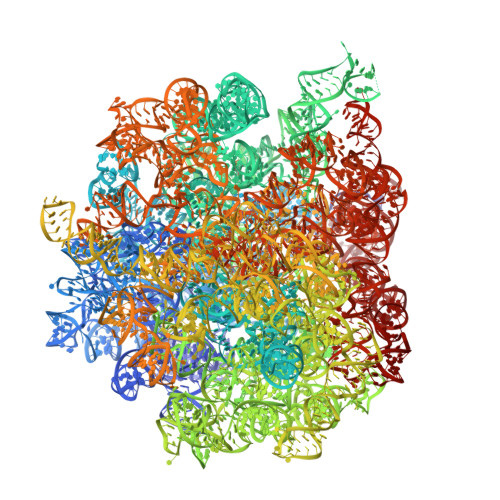
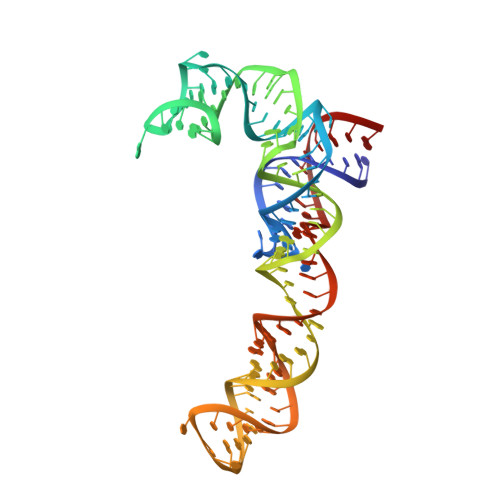
Structure of Escherichia coli heat shock protein Hsp15 in complex with the ribosomal 50S subunit bearing peptidyl-tRNA.
Safdari, H.A., Kasvandik, S., Polte, C., Ignatova, Z., Tenson, T., Wilson, D.N.(2022) Nucleic Acids Res 50: 12515-12526
- PubMed: 36370110 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkac1035
- Primary Citation Related Structures:
8AP4 - PubMed Abstract:
In Escherichia coli, the heat shock protein 15 (Hsp15) is part of the cellular response to elevated temperature. Hsp15 interacts with peptidyl-tRNA-50S complexes that arise upon dissociation of translating 70S ribosomes, and is proposed to facilitate their rescue and recycling. A previous structure of E. coli Hsp15 in complex with peptidyl-tRNA-50S complex reported a binding site located at the central protuberance of the 50S subunit. By contrast, recent structures of RqcP, the Hsp15 homolog in Bacillus subtilis, in complex with peptidyl-tRNA-50S complexes have revealed a distinct site positioned between the anticodon-stem-loop (ASL) of the P-site tRNA and H69 of the 23S rRNA. Here we demonstrate that exposure of E. coli cells to heat shock leads to a decrease in 70S ribosomes and accumulation of 50S subunits, thus identifying a natural substrate for Hsp15 binding. Additionally, we have determined a cryo-EM reconstruction of the Hsp15-50S-peptidyl-tRNA complex isolated from heat shocked E. coli cells, revealing that Hsp15 binds to the 50S-peptidyl-tRNA complex analogously to its B. subtilis homolog RqcP. Collectively, our findings support a model where Hsp15 stabilizes the peptidyl-tRNA in the P-site and thereby promotes access to the A-site for putative rescue factors to release the aberrant nascent polypeptide chain.
- Institute for Biochemistry and Molecular Biology, University of Hamburg, Martin-Luther-King-Pl. 6, 20146 Hamburg, Germany.
Organizational Affiliation: