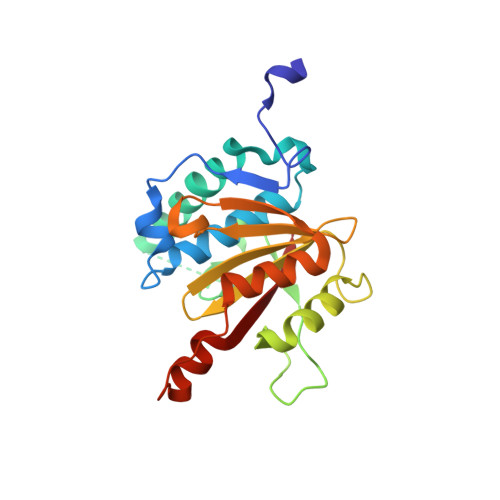
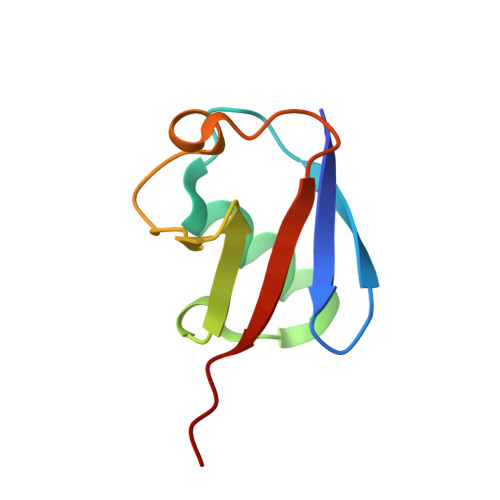
A widely distributed family of eukaryotic and bacterial deubiquitinases related to herpesviral large tegument proteins.
Erven, I., Abraham, E., Hermanns, T., Baumann, U., Hofmann, K.(2022) Nat Commun 13: 7643-7643
- PubMed: 36496440 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-35244-y
- Primary Citation Related Structures:
8ADB, 8ADC, 8ADD - PubMed Abstract:
Distinct families of eukaryotic deubiquitinases (DUBs) are regulators of ubiquitin signaling. Here, we report on the presence of an additional DUB class broadly distributed in eukaryotes and several bacteria. The only described members of this family are the large tegument proteins of herpesviruses, which are attached to the outside of the viral capsid. By using a bioinformatics screen, we have identified distant homologs of this VTD (Viral tegument-like DUB) family in vertebrate transposons, fungi, insects, nematodes, cnidaria, protists and bacteria. While some VTD activities resemble viral tegument DUBs in that they favor K48-linked ubiquitin chains, other members are highly specific for K6- or K63-linked ubiquitin chains. The crystal structures of K48- and K6-specific members reveal considerable differences in ubiquitin recognition. The VTD family likely evolved from non-DUB proteases and spread through transposons, many of which became 'domesticated', giving rise to the Drosophila male sterile (3)76Ca gene and several nematode genes with male-specific expression.
- Institute for Genetics, University of Cologne, Zülpicher Straße 47a, D-50674, Cologne, Germany.
Organizational Affiliation: