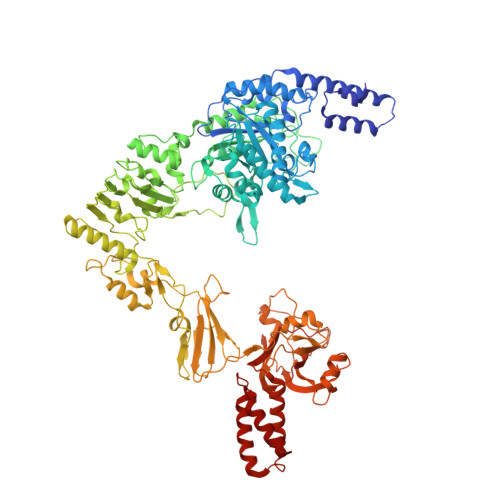
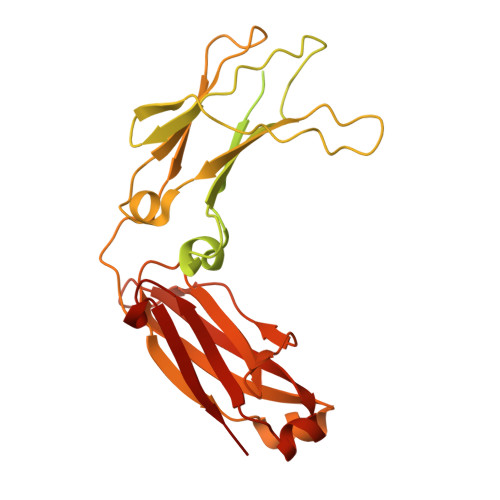
Mechanism of antibody-specific deglycosylation and immune evasion by Streptococcal IgG-specific endoglycosidases.
Trastoy, B., Du, J.J., Cifuente, J.O., Rudolph, L., Garcia-Alija, M., Klontz, E.H., Deredge, D., Sultana, N., Huynh, C.G., Flowers, M.W., Li, C., Sastre, D.E., Wang, L.X., Corzana, F., Mallagaray, A., Sundberg, E.J., Guerin, M.E.(2023) Nat Commun 14: 1705-1705
- PubMed: 36973249 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-023-37215-3
- Primary Citation Related Structures:
8A64 - PubMed Abstract:
Bacterial pathogens have evolved intricate mechanisms to evade the human immune system, including the production of immunomodulatory enzymes. Streptococcus pyogenes serotypes secrete two multi-modular endo-β-N-acetylglucosaminidases, EndoS and EndoS2, that specifically deglycosylate the conserved N-glycan at Asn297 on IgG Fc, disabling antibody-mediated effector functions. Amongst thousands of known carbohydrate-active enzymes, EndoS and EndoS2 represent just a handful of enzymes that are specific to the protein portion of the glycoprotein substrate, not just the glycan component. Here, we present the cryoEM structure of EndoS in complex with the IgG1 Fc fragment. In combination with small-angle X-ray scattering, alanine scanning mutagenesis, hydrolytic activity measurements, enzyme kinetics, nuclear magnetic resonance and molecular dynamics analyses, we establish the mechanisms of recognition and specific deglycosylation of IgG antibodies by EndoS and EndoS2. Our results provide a rational basis from which to engineer novel enzymes with antibody and glycan selectivity for clinical and biotechnological applications.
- Structural Glycobiology Laboratory, Biocruces Health Research Institute, Barakaldo, Bizkaia, 48903, Spain. beatriz.trastoy@gmail.com.
Organizational Affiliation: