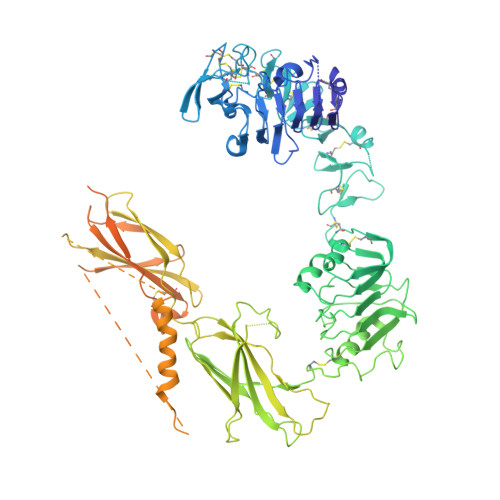
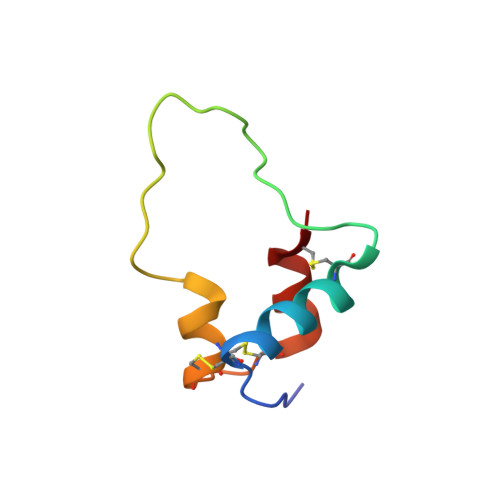
Interaction of a viral insulin-like peptide with the IGF-1 receptor produces a natural antagonist.
Moreau, F., Kirk, N.S., Zhang, F., Gelfanov, V., List, E.O., Chrudinova, M., Venugopal, H., Lawrence, M.C., Jimenez, V., Bosch, F., Kopchick, J.J., DiMarchi, R.D., Altindis, E., Ronald Kahn, C.(2022) Nat Commun 13: 6700-6700
- PubMed: 36335114 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-34391-6
- Primary Citation Related Structures:
7U23 - PubMed Abstract:
Lymphocystis disease virus-1 (LCDV-1) and several other Iridoviridae encode viral insulin/IGF-1 like peptides (VILPs) with high homology to human insulin and IGFs. Here we show that while single-chain (sc) and double-chain (dc) LCDV1-VILPs have very low affinity for the insulin receptor, scLCDV1-VILP has high affinity for IGF1R where it can antagonize human IGF-1 signaling, without altering insulin signaling. Consequently, scLCDV1-VILP inhibits IGF-1 induced cell proliferation and growth hormone/IGF-1 induced growth of mice in vivo. Cryo-electron microscopy reveals that scLCDV1-VILP engages IGF1R in a unique manner, inducing changes in IGF1R conformation that led to separation, rather than juxtaposition, of the transmembrane segments and hence inactivation of the receptor. Thus, scLCDV1-VILP is a natural peptide with specific antagonist properties on IGF1R signaling and may provide a new tool to guide development of hormonal analogues to treat cancers or metabolic disorders sensitive to IGF-1 without affecting glucose metabolism.
- Section of Integrative Physiology and Metabolism, Joslin Diabetes Center, Harvard Medical School, Boston, MA, USA.
Organizational Affiliation: