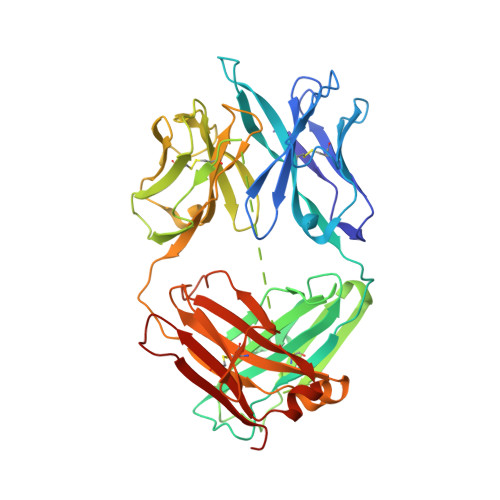
CAR T cells targeting tumor-associated exons of glypican 2 regress neuroblastoma in mice.
Li, N., Torres, M.B., Spetz, M.R., Wang, R., Peng, L., Tian, M., Dower, C.M., Nguyen, R., Sun, M., Tai, C.H., de Val, N., Cachau, R., Wu, X., Hewitt, S.M., Kaplan, R.N., Khan, J., St Croix, B., Thiele, C.J., Ho, M.(2021) Cell Rep Med 2: 100297-100297
- PubMed: 34195677 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.xcrm.2021.100297
- Primary Citation Related Structures:
7T62 - PubMed Abstract:
Targeting solid tumors must overcome several major obstacles, in particular, the identification of elusive tumor-specific antigens. Here, we devise a strategy to help identify tumor-specific epitopes. Glypican 2 (GPC2) is overexpressed in neuroblastoma. Using RNA sequencing (RNA-seq) analysis, we show that exon 3 and exons 7-10 of GPC2 are expressed in cancer but are minimally expressed in normal tissues. Accordingly, we discover a monoclonal antibody (CT3) that binds exons 3 and 10 and visualize the complex structure of CT3 and GPC2 by electron microscopy. The potential of this approach is exemplified by designing CT3-derived chimeric antigen receptor (CAR) T cells that regress neuroblastoma in mice. Genomic sequencing of T cells recovered from mice reveals the CAR integration sites that may contribute to CAR T cell proliferation and persistence. These studies demonstrate how RNA-seq data can be exploited to help identify tumor-associated exons that can be targeted by CAR T cell therapies.
- Laboratory of Molecular Biology, Center for Cancer Research, National Cancer Institute, National Institutes of Health, Bethesda, MD 20892, USA.
Organizational Affiliation: