Structural basis for the metabolism of xenobiotic C-glycosides by intestinal bacteria
Mori, T., Kumano, T., He, K., Awakawa, T., Watanabe, S., Hori, S., Terashita, Y., Hashimoto, Y., Senda, M., Senda, T., Kobayashi, M., Abe, I.(2021) Nat Commun
Experimental Data Snapshot
(2021) Nat Commun
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| AP_endonuc_2 domain-containing protein | A, C [auth P] | 354 | Arthrobacter globiformis NBRC 12137 | Mutation(s): 0 Gene Names: ARGLB_075_00530 EC: 4.1.99 | 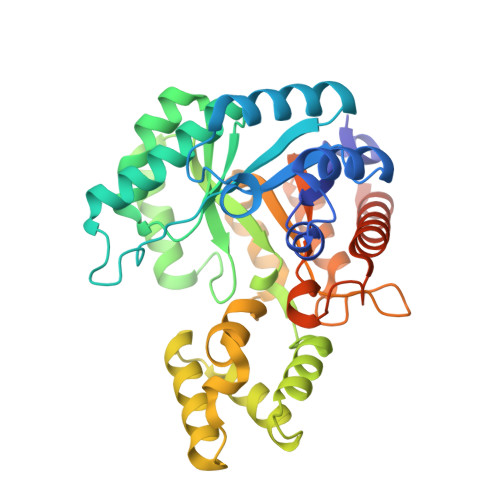 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | H0QPL9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| AgCarC2 | B, D [auth Q] | 145 | Arthrobacter globiformis NBRC 12137 | Mutation(s): 0 Gene Names: ARGLB_075_00520 EC: 4.1.99 | 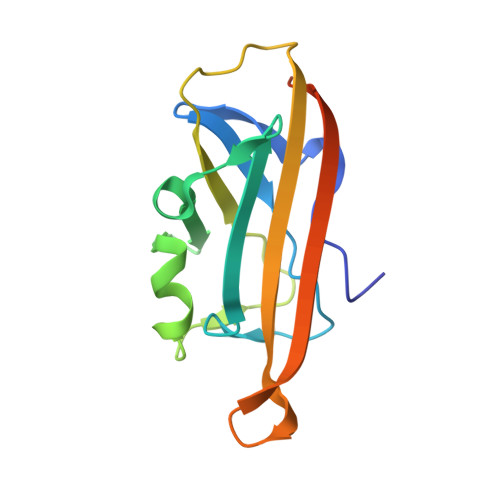 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | H0QPL8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| H9R (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | E [auth A] | 2-[3,4-bis(oxidanyl)phenyl]-6-[(2S,3R,4R,5S,6R)-6-(hydroxymethyl)-3,4,5-tris(oxidanyl)oxan-2-yl]-5,7-bis(oxidanyl)chromen-4-one C21 H20 O11 ODBRNZZJSYPIDI-VJXVFPJBSA-N |  | ||
| IOD Download:Ideal Coordinates CCD File | G [auth A], I [auth P] | IODIDE ION I XMBWDFGMSWQBCA-UHFFFAOYSA-M |  | ||
| MN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | F [auth A], H [auth P] | MANGANESE (II) ION Mn WAEMQWOKJMHJLA-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 73.905 | α = 90 |
| b = 102.511 | β = 90 |
| c = 136.09 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |
| CRANK2 | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Japan Agency for Medical Research and Development (AMED) | Japan | JP19am0101001 |