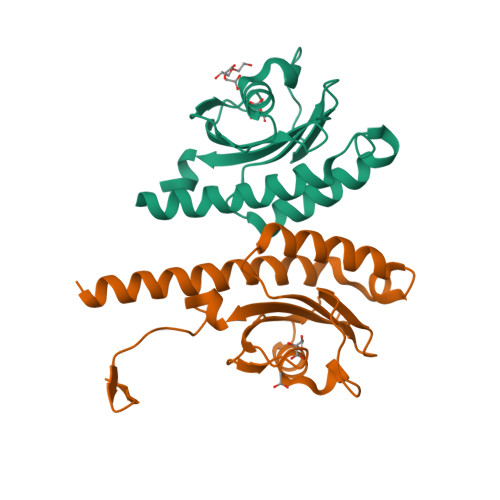
Signal binding at both modules of its dCache domain enables the McpA chemoreceptor of Bacillus velezensis to sense different ligands.
Feng, H., Lv, Y., Krell, T., Fu, R., Liu, Y., Xu, Z., Du, W., Shen, Q., Zhang, N., Zhang, R.(2022) Proc Natl Acad Sci U S A 119: e2201747119-e2201747119
- PubMed: 35858353
- DOI: https://doi.org/10.1073/pnas.2201747119
- Primary Citation of Related Structures:
7W0W - PubMed Abstract:
Bacteria have evolved multiple signal transduction systems that permit an adaptation to changing environmental conditions. Chemoreceptor-based signaling cascades are very abundant in bacteria and are among the most complex signaling systems. Currently, our knowledge on the molecular features that determine signal recognition at chemoreceptors is limited. Chemoreceptor McpA of Bacillus velezensis SQR9 has been shown to mediate chemotaxis to a broad range of different ligands. Here we show that its ligand binding domain binds directly 13 chemoattractants. We provide support that organic acids and amino acids bind to the membrane-distal and membrane-proximal module of the dCache domain, respectively, whereas binding of sugars/sugar alcohols occurred at both modules. Structural biology studies combined with site-directed mutagenesis experiments have permitted to identify 10 amino acid residues that play key roles in the recognition of multiple ligands. Residues in membrane-distal and membrane-proximal regions were central for sensing organic acids and amimo acids, respectively, whereas all residues participated in sugars/sugar alcohol sensing. Most characterized chemoreceptors possess a narrow and well-defined ligand spectrum. We propose here a sensing mechanism involving both dCache modules that allows the integration of very diverse signals by a single chemoreceptor.
Organizational Affiliation:
Jiangsu Provincial Key Lab of Solid Organic Waste Utilization, Jiangsu Collaborative Innovation Center of Solid Organic Wastes, Educational Ministry Engineering Center of Resource-Saving Fertilizers, The Key Laboratory of Plant Immunity, Nanjing Agricultural University, 210095 Nanjing, People's Republic of China.