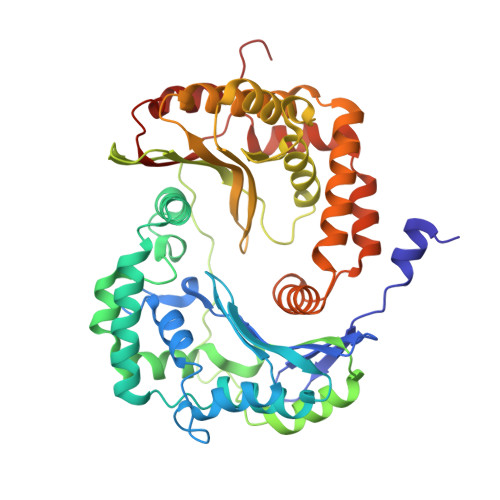
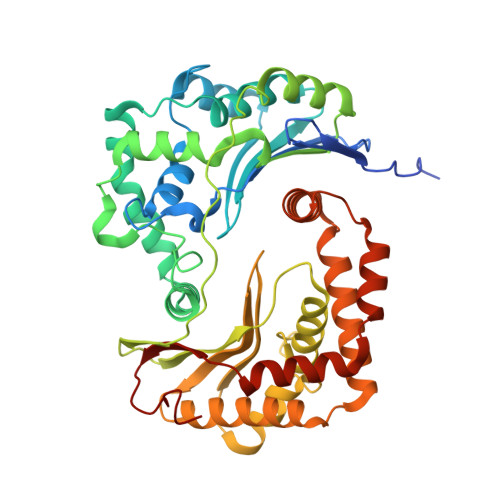
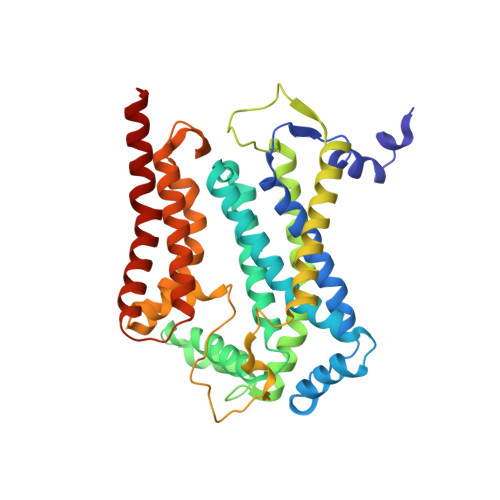
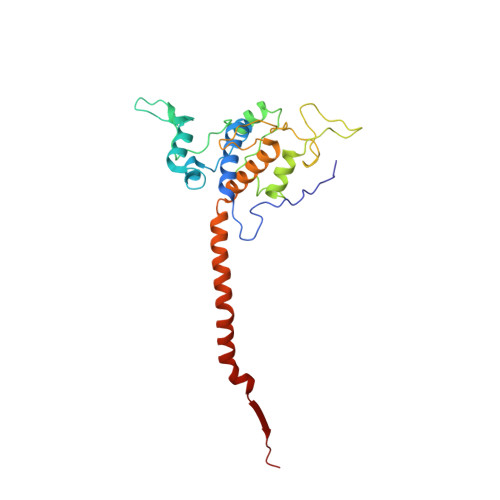
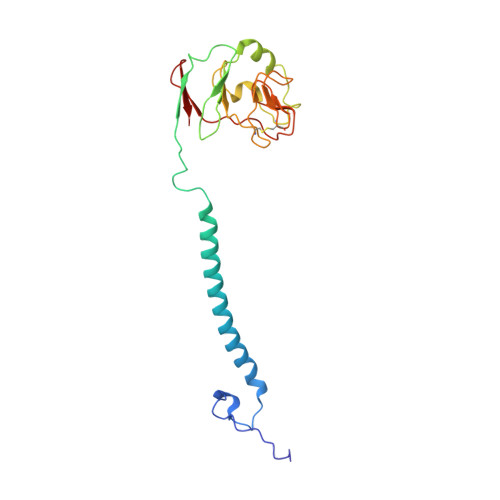
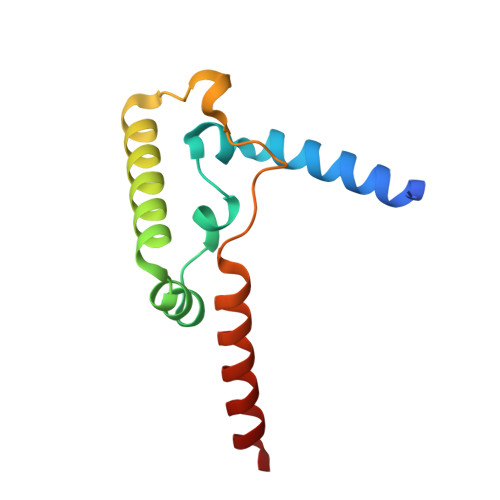
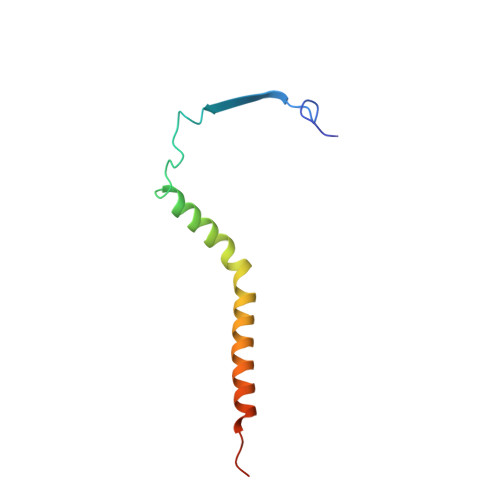
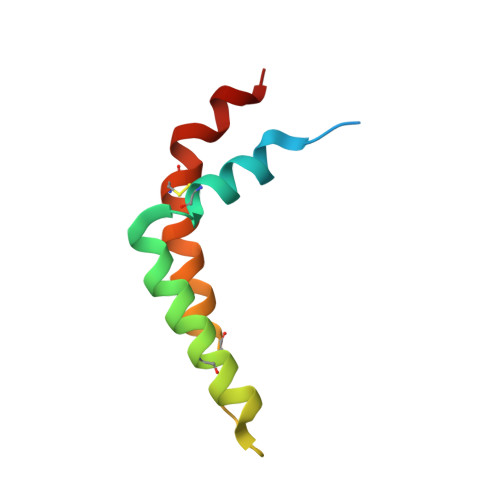
Structure of complex III with bound antimalarial agent CK-2-68 provides insights into selective inhibition of Plasmodium cytochrome bc 1 complexes.
Esser, L., Zhou, F., Zeher, A., Wu, W., Huang, R., Yu, C.A., Lane, K.D., Wellems, T.E., Xia, D.(2023) J Biological Chem 299: 104860-104860
- PubMed: 37236355 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2023.104860
- Primary Citation Related Structures:
7TZ6 - PubMed Abstract:
Among the various components of the protozoan Plasmodium mitochondrial respiratory chain, only Complex III is a validated cellular target for antimalarial drugs. The compound CK-2-68 was developed to specifically target the alternate NADH dehydrogenase of the malaria parasite respiratory chain, but the true target for its antimalarial activity has been controversial. Here, we report the cryo-EM structure of mammalian mitochondrial Complex III bound with CK-2-68 and examine the structure-function relationships of the inhibitor's selective action on Plasmodium. We show that CK-2-68 binds specifically to the quinol oxidation site of Complex III, arresting the motion of the iron-sulfur protein subunit, which suggests an inhibition mechanism similar to that of P f -type Complex III inhibitors such as atovaquone, stigmatellin, and UHDBT. Our results shed light on the mechanisms of observed resistance conferred by mutations, elucidate the molecular basis of the wide therapeutic window of CK-2-68 for selective action of Plasmodium vs. host cytochrome bc 1 , and provide guidance for future development of antimalarials targeting Complex III.
- Laboratory of Cell Biology, Center for Cancer Research National Cancer Institute, NIH, Bethesda, Maryland, USA.
Organizational Affiliation: