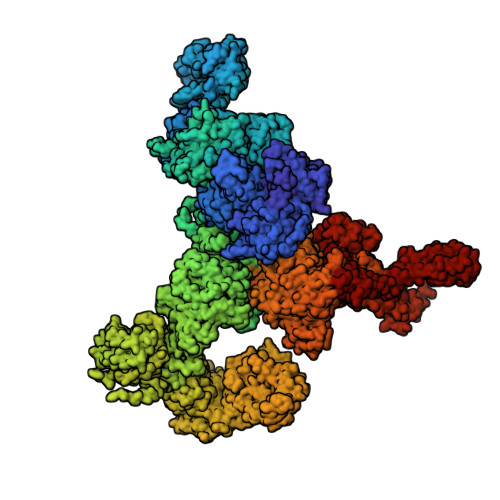
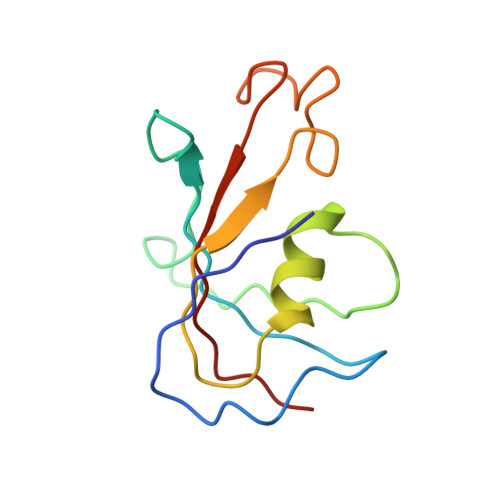
Molecular mechanism of the severe MH/CCD mutation Y522S in skeletal ryanodine receptor (RyR1) by cryo-EM.
Iyer, K.A., Hu, Y., Klose, T., Murayama, T., Samso, M.(2022) Proc Natl Acad Sci U S A 119: e2122140119-e2122140119
- PubMed: 35867837 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.2122140119
- Primary Citation Related Structures:
7T64, 7T65 - PubMed Abstract:
Ryanodine receptors (RyRs) are main regulators of intracellular Ca 2+ release and muscle contraction. The Y522S mutation of RyR1 causes central core disease, a weakening myopathy, and malignant hyperthermia, a sudden and potentially fatal response to anesthetics or heat. Y522 is in the core of the N-terminal subdomain C of RyR1 and the mechanism of how this mutation orchestrates malfunction is unpredictable for this 2-MDa ion channel, which has four identical subunits composed of 15 distinct cytoplasmic domains each. We expressed and purified the RyR1 rabbit homolog, Y523S, from HEK293 cells and reconstituted it in nanodiscs under closed and open states. The high-resolution cryogenic electron microscopic (cryo-EM) three-dimensional (3D) structures show that the phenyl ring of Tyr functions in a manner analogous to a "spacer" within an α-helical bundle. Mutation to the much smaller Ser alters the hydrophobic network within the bundle, triggering rearrangement of its α-helices with repercussions in the orientation of most cytoplasmic domains. Examining the mutation-induced readjustments exposed a series of connected α-helices acting as an ∼100 Å-long lever: One end protrudes toward the dihydropyridine receptor, its molecular activator (akin to an antenna), while the other end reaches the Ca 2+ activation site. The Y523S mutation elicits channel preactivation in the absence of any activator and full opening at 1.5 µM free Ca 2+ , increasing by ∼20-fold the potency of Ca 2+ to activate the channel compared with RyR1 wild type (WT). This study identified a preactivated pathological state of RyR1 and a long-range lever that may work as a molecular switch to open the channel.
- Department of Physiology and Biophysics, School of Medicine, Virginia Commonwealth University, Richmond, VA 23298.
Organizational Affiliation: