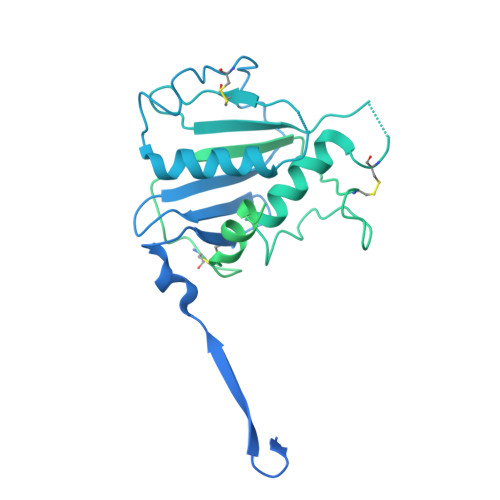
Lassa virus glycoprotein nanoparticles elicit neutralizing antibody responses and protection.
Brouwer, P.J.M., Antanasijevic, A., Ronk, A.J., Muller-Krauter, H., Watanabe, Y., Claireaux, M., Perrett, H.R., Bijl, T.P.L., Grobben, M., Umotoy, J.C., Schriek, A.I., Burger, J.A., Tejjani, K., Lloyd, N.M., Steijaert, T.H., van Haaren, M.M., Sliepen, K., de Taeye, S.W., van Gils, M.J., Crispin, M., Strecker, T., Bukreyev, A., Ward, A.B., Sanders, R.W.(2022) Cell Host Microbe 30: 1759
- PubMed: 36400021 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.chom.2022.10.018
- Primary Citation Related Structures:
7SGD, 7SGE, 7SGF - PubMed Abstract:
The Lassa virus is endemic in parts of West Africa, and it causes hemorrhagic fever with high mortality. The development of a recombinant protein vaccine has been hampered by the instability of soluble Lassa virus glycoprotein complex (GPC) trimers, which disassemble into monomeric subunits after expression. Here, we use two-component protein nanoparticles consisting of trimeric and pentameric subunits to stabilize GPC in a trimeric conformation. These GPC nanoparticles present twenty prefusion GPC trimers on the surface of an icosahedral particle. Cryo-EM studies of GPC nanoparticles demonstrated a well-ordered structure and yielded a high-resolution structure of an unliganded GPC. These nanoparticles induced potent humoral immune responses in rabbits and protective immunity against the lethal Lassa virus challenge in guinea pigs. Additionally, we isolated a neutralizing antibody that mapped to the putative receptor-binding site, revealing a previously undefined site of vulnerability. Collectively, these findings offer potential approaches to vaccine and therapeutic design for the Lassa virus.
- Department of Medical Microbiology and Infection Prevention, Amsterdam University Medical Centers, Location AMC, University of Amsterdam, Amsterdam Infection & Immunity Institute, 1105 AZ Amsterdam, the Netherlands; Department of Integrative, Structural and Computational Biology, The Scripps Research Institute, La Jolla, CA 92037, USA.
Organizational Affiliation: