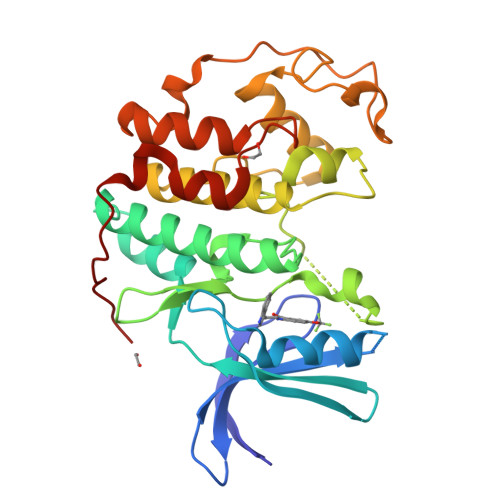
Screening through Lead Optimization of High Affinity, Allosteric Cyclin-Dependent Kinase 2 (CDK2) Inhibitors as Male Contraceptives That Reduce Sperm Counts in Mice.
Faber, E.B., Wang, N., John, K., Sun, L., Wong, H.L., Burban, D., Francis, R., Tian, D., Hong, K.H., Yang, A., Wang, L., Elsaid, M., Khalid, H., Levinson, N.M., Schonbrunn, E., Hawkinson, J.E., Georg, G.I.(2023) J Med Chem 66: 1928-1940
- PubMed: 36701569
- DOI: https://doi.org/10.1021/acs.jmedchem.2c01731
- Primary Citation of Related Structures:
7RWE, 7RXO, 7S4T, 7S7A, 7S85 - PubMed Abstract:
Although cyclin-dependent kinase 2 (CDK2) is a validated target for both cancer and contraception, developing a CDK2 inhibitor with exquisite selectivity has been challenging due to the structural similarity of the ATP-binding site, where most kinase inhibitors bind. We previously discovered an allosteric pocket in CDK2 with the potential to bind a selective compound and then discovered and structurally confirmed an anthranilic acid scaffold that binds this pocket with high affinity. These allosteric inhibitors are selective for CDK2 over structurally similar CDK1 and show contraceptive potential. Herein, we describe the screening and optimization that led to compounds like EF-4-177 with nanomolar affinity for CDK2. EF-4-177 is metabolically stable, orally bioavailable, and significantly disrupts spermatogenesis, demonstrating this series' therapeutic potential. This work details the discovery of the highest affinity allosteric CDK inhibitors reported and shows promise for this series to yield an efficacious and selective allosteric CDK2 inhibitor.
Organizational Affiliation:
Department of Medicinal Chemistry and Institute for Therapeutics Discovery and Development, University of Minnesota College of Pharmacy─Twin Cities, Minneapolis, Minnesota55414, United States.