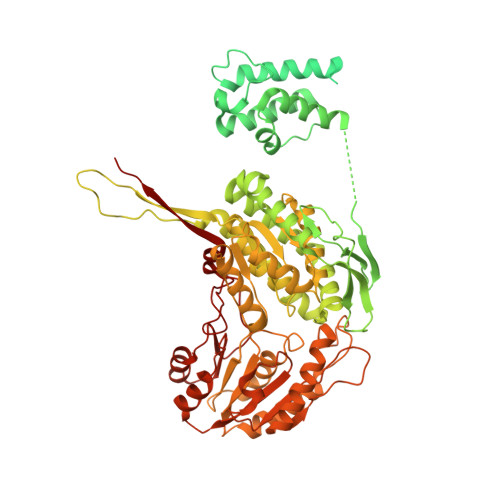
Structure of putative tumor suppressor ALDH1L1.
Tsybovsky, Y., Sereda, V., Golczak, M., Krupenko, N.I., Krupenko, S.A.(2022) Commun Biol 5: 3-3
- PubMed: 35013550 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s42003-021-02963-9
- PubMed Abstract:
Putative tumor suppressor ALDH1L1, the product of natural fusion of three unrelated genes, regulates folate metabolism by catalyzing NADP + -dependent conversion of 10-formyltetrahydrofolate to tetrahydrofolate and CO 2 . Cryo-EM structures of tetrameric rat ALDH1L1 revealed the architecture and functional domain interactions of this complex enzyme. Highly mobile N-terminal domains, which remove formyl from 10-formyltetrahydrofolate, undergo multiple transient inter-domain interactions. The C-terminal aldehyde dehydrogenase domains, which convert formyl to CO 2 , form unusually large interfaces with the intermediate domains, homologs of acyl/peptidyl carrier proteins (A/PCPs), which transfer the formyl group between the catalytic domains. The 4'-phosphopantetheine arm of the intermediate domain is fully extended and reaches deep into the catalytic pocket of the C-terminal domain. Remarkably, the tetrameric state of ALDH1L1 is indispensable for catalysis because the intermediate domain transfers formyl between the catalytic domains of different protomers. These findings emphasize the versatility of A/PCPs in complex, highly dynamic enzymatic systems.
- Cancer Research Technology Program, Leidos Biomedical Research Inc., Frederick National Laboratory for Cancer Research, 8560 Progress Drive, Frederick, MD, 21701, USA. Yaroslav.Tsybovsky@nih.gov.
Organizational Affiliation: