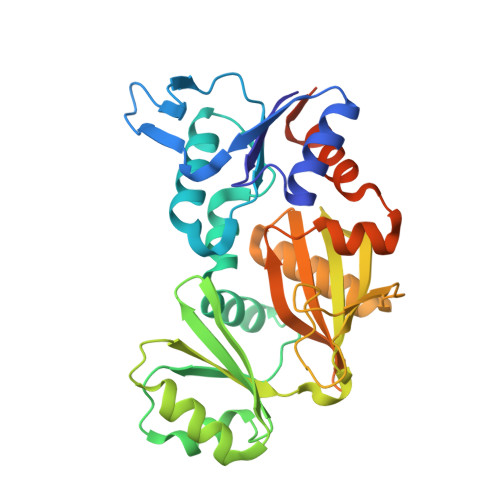
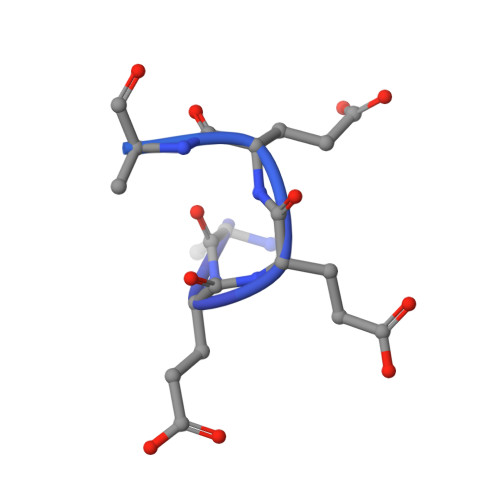
Structural insights into the mechanism of adaptive ribosomal modification by Pseudomonas RimK.
Thompson, C.M.A., Little, R.H., Stevenson, C.E.M., Lawson, D.M., Malone, J.G.(2023) Proteins 91: 300-314
- PubMed: 36134899 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/prot.26429
- Primary Citation Related Structures:
7QYR, 7QYS - PubMed Abstract:
Bacteria are equipped with a diverse set of regulatory tools that allow them to quickly adapt to their environment. The RimK system allows for Pseudomonas spp. to adapt through post-transcriptional regulation by altering the ribosomal subunit RpsF. RimK is found in a wide range of bacteria with a conserved amino acid sequence, however, the genetic context and the role of this protein is highly diverse. By solving and comparing the structures of RimK homologs from two related but functionally divergent systems, we uncovered key structural differences that likely contribute to the different activity levels of each of these homologs. Moreover, we were able to clearly resolve the active site of this protein for the first time, resolving binding of the glutamate substrate. This work advances our understanding of how subtle differences in protein sequence and structure can have profound effects on protein activity, which can in turn result in widespread mechanistic changes.
- Department of Molecular Microbiology, John Innes Centre, Norwich Research Park, Norwich, United Kingdom.
Organizational Affiliation: