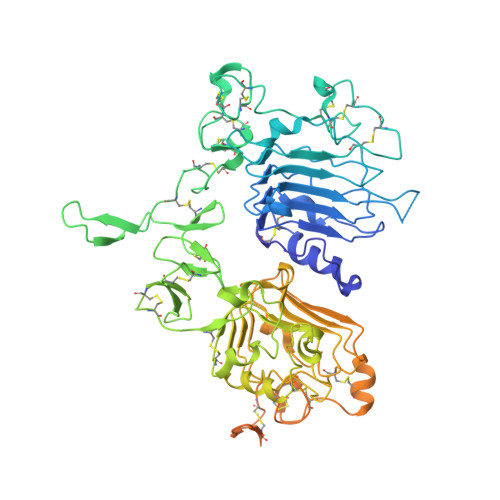
Co-crystallisation and humanisation of an anti-HER2 single-domain antibody as a theranostic tool.
Sawmynaden, K., Wong, N., Davies, S., Cowan, R., Brown, R., Tang, D., Henry, M., Tickle, D., Matthews, D., Carr, M., Bakrania, P., Hoi Ting, H., Hall, G.(2023) PLoS One 18: e0288259-e0288259
- PubMed: 37459326 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0288259
- Primary Citation Related Structures:
7QVK - PubMed Abstract:
Human epidermal growth factor receptor-2 (HER2) is a well-recognised biomarker associated with 25% of breast cancers. In most cases, early detection and/or treatment correlates with an increased chance of survival. This study, has identified and characterised a highly specific anti-HER2 single-domain antibody (sdAb), NM-02, as a potential theranostic tool. Complete structural description by X-ray crystallography has revealed a non-overlapping epitope with current anti-HER2 antibodies. To reduce the immunogenicity risk, NM-02 underwent a humanisation process and retained wild type-like binding properties. To further de-risk the progression towards chemistry, manufacturing and control (CMC) we performed full developability profiling revealing favourable thermal and physical biochemical 'drug-like' properties. Finally, the application of the lead humanised NM-02 candidate (variant K) for HER2-specific imaging purposes was demonstrated using breast cancer HER2+/BT474 xenograft mice.
- LifeArc, Open Innovation Campus, Stevenage, United Kingdom.
Organizational Affiliation: