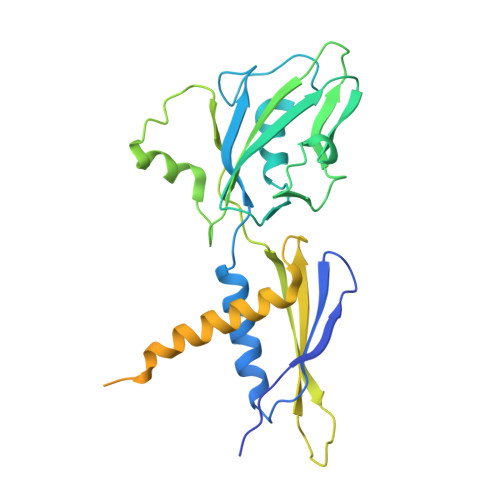
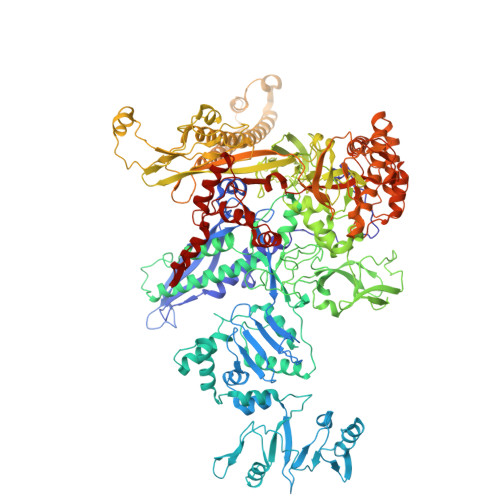
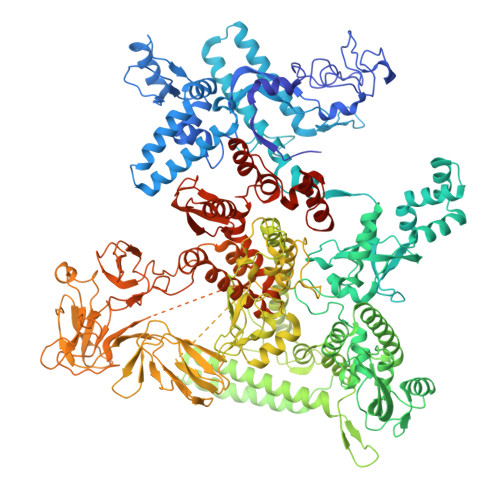
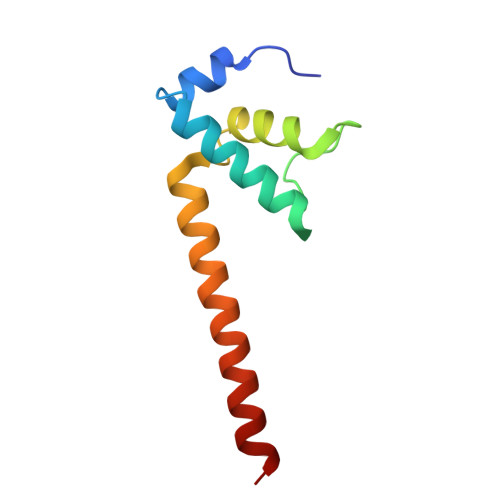
Transcription factors modulate RNA polymerase conformational equilibrium.
Zhu, C., Guo, X., Dumas, P., Takacs, M., Abdelkareem, M., Vanden Broeck, A., Saint-Andre, C., Papai, G., Crucifix, C., Ortiz, J., Weixlbaumer, A.(2022) Nat Commun 13: 1546-1546
- PubMed: 35318334 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/s41467-022-29148-0
- Primary Citation Related Structures:
7PY0, 7PY1, 7PY3, 7PY5, 7PY6, 7PY7, 7PY8, 7PYJ, 7PYK, 7Q0J, 7Q0K - PubMed Abstract:
RNA polymerase (RNAP) frequently pauses during the transcription of DNA to RNA to regulate gene expression. Transcription factors NusA and NusG modulate pausing, have opposing roles, but can bind RNAP simultaneously. Here we report cryo-EM reconstructions of Escherichia coli RNAP bound to NusG, or NusA, or both. RNAP conformational changes, referred to as swivelling, correlate with transcriptional pausing. NusA facilitates RNAP swivelling to further increase pausing, while NusG counteracts this role. Their structural effects are consistent with biochemical results on two categories of transcriptional pauses. In addition, the structures suggest a cooperative mechanism of NusA and NusG during Rho-mediated transcription termination. Our results provide a structural rationale for the stochastic nature of pausing and termination and how NusA and NusG can modulate it.
- Department of Integrated Structural Biology, Institut de Génétique et de Biologie Moléculaire et Cellulaire (IGBMC), 67404, Illkirch, France.
Organizational Affiliation: