Cryo-EM structure of the human TRPA1 ion channel in complex with the antagonist 3-60
Grieben, M., Saward, B.G., Pike, A.C.W., Schofield, C.J., Carpenter, E.P.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Transient receptor potential cation channel subfamily A member 1 | 1,126 | Homo sapiens | Mutation(s): 2 Gene Names: TRPA1, ANKTM1 Membrane Entity: Yes | 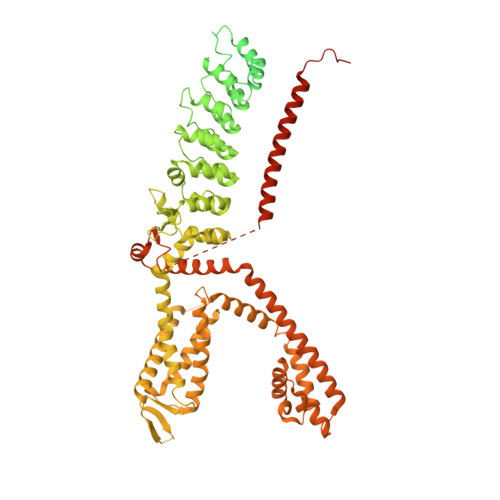 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: O75762 GTEx: ENSG00000104321 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O75762 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | Go to GlyGen: O75762-1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PC1 Download:Ideal Coordinates CCD File | DA [auth D] EA [auth D] J [auth A] K [auth A] P [auth B] | 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE C44 H88 N O8 P NRJAVPSFFCBXDT-HUESYALOSA-N |  | ||
| IHP Download:Ideal Coordinates CCD File | AA [auth C], BA [auth D], N [auth A], T [auth B] | INOSITOL HEXAKISPHOSPHATE C6 H18 O24 P6 IMQLKJBTEOYOSI-GPIVLXJGSA-N |  | ||
| 0IG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | CA [auth D], I [auth A], O [auth B], V [auth C] | ~{N}-[4-[4-[(azanylidene-$l^{4}-azanylidene)amino]phenyl]-1,3-thiazol-2-yl]-2-[1,3-dimethyl-2,6-bis(oxidanylidene)purin-7-yl]ethanamide C18 H16 N9 O3 S BCMLFCZGHWVERY-UHFFFAOYSA-O |  | ||
| NAG Download:Ideal Coordinates CCD File | GA [auth D], M [auth A], S [auth B], Z [auth C] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | FA [auth D], L [auth A], R [auth B], U [auth B], Y [auth C] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | cryoSPARC |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Wellcome Trust | United Kingdom | 106169/Z/14/Z |
| European Commission | United Kingdom | 115766 |