E3 ubiquitin ligase RNF213 employs a non-canonical zinc finger active site and is allosterically regulated by ATP
Ahel, J., Fletcher, A.J., Grabarczyk, D., Roitinger, E., Deszcz, L., Lehner, A., Virdee, S., Clausen, T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| E3 ubiquitin-protein ligase RNF213 | 4,836 | Mus musculus | Mutation(s): 0 Gene Names: Rnf213, Mystr EC: 2.3.2.27 (PDB Primary Data), 3.6.4 (PDB Primary Data), 2.3.2 (UniProt) | 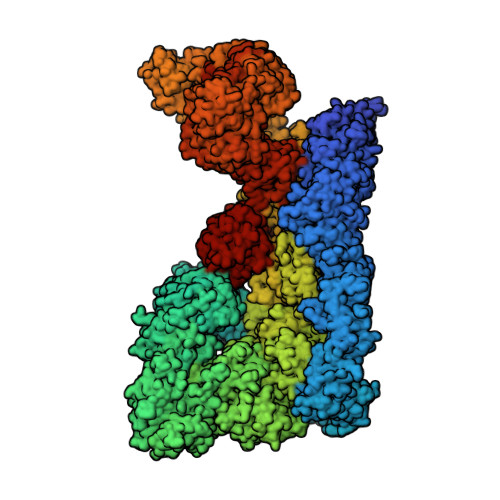 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | E9Q555 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| AGS (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | G [auth A] | PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER C10 H16 N5 O12 P3 S NLTUCYMLOPLUHL-KQYNXXCUSA-N |  | ||
| ATP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | B [auth A] | ADENOSINE-5'-TRIPHOSPHATE C10 H16 N5 O13 P3 ZKHQWZAMYRWXGA-KQYNXXCUSA-N |  | ||
| ADP (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | F [auth A] | ADENOSINE-5'-DIPHOSPHATE C10 H15 N5 O10 P2 XTWYTFMLZFPYCI-KQYNXXCUSA-N |  | ||
| ZN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth A], E [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| MG (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | C [auth A] | MAGNESIUM ION Mg JLVVSXFLKOJNIY-UHFFFAOYSA-N |  | ||
| Task | Software Package | Version |
|---|---|---|
| RECONSTRUCTION | RELION | 3.1 |
| MODEL REFINEMENT | PHENIX |
| Funding Organization | Location | Grant Number |
|---|---|---|
| European Research Council (ERC) | Austria | 694978 |
| Austrian Research Promotion Agency | Austria | 852936 |
| Medical Research Council (MRC, United Kingdom) | United Kingdom | MC_UU_12016/8 |
| Biotechnology and Biological Sciences Research Council (BBSRC) | United Kingdom | BB/ P003982/1 |