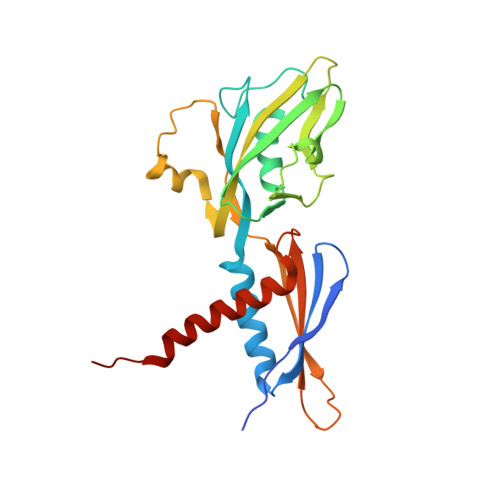
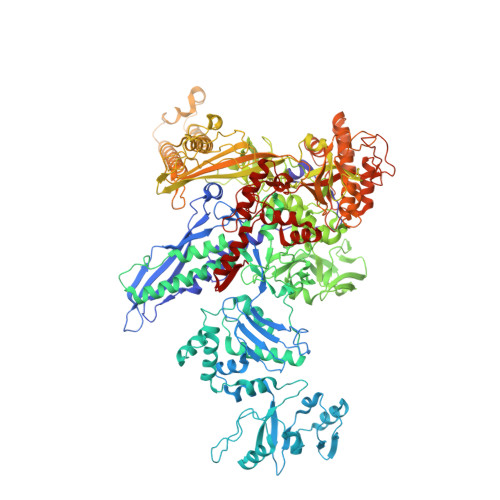
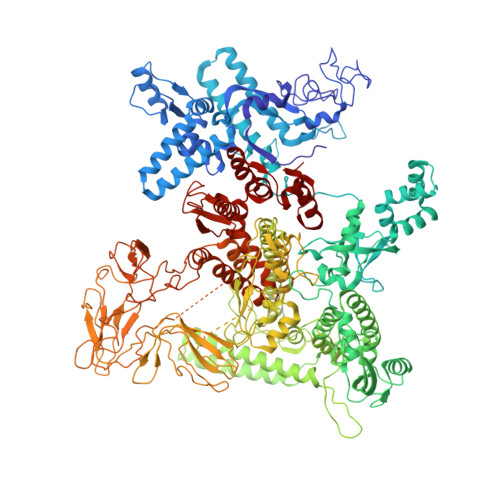
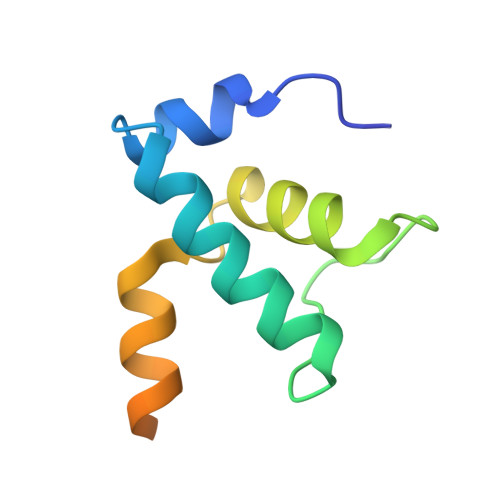
Structural basis of RNA polymerase recycling by the Swi2/Snf2 family of ATPase RapA in Escherichia coli.
Qayyum, M.Z., Molodtsov, V., Renda, A., Murakami, K.S.(2021) J Biological Chem 297: 101404-101404
- PubMed: 34774797 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jbc.2021.101404
- Primary Citation Related Structures:
7MKN, 7MKO, 7MKP, 7MKQ - PubMed Abstract:
After transcription termination, cellular RNA polymerases (RNAPs) are occasionally trapped on DNA, impounded in an undefined post-termination complex (PTC), limiting the free RNAP pool and subsequently leading to inefficient transcription. In Escherichia coli, a Swi2/Snf2 family of ATPase called RapA is known to be involved in countering such inefficiency through RNAP recycling; however, the precise mechanism of this recycling is unclear. To better understand its mechanism, here we determined the structures of two sets of E. coli RapA-RNAP complexes, along with the RNAP core enzyme and the elongation complex, using cryo-EM. These structures revealed the large conformational changes of RNAP and RapA upon their association that has been implicated in the hindrance of PTC formation. Our results along with DNA-binding assays reveal that although RapA binds RNAP away from the DNA-binding main channel, its binding can allosterically close the RNAP clamp, thereby preventing its nonspecific DNA binding and PTC formation. Taken together, we propose that RapA acts as a guardian of RNAP by which RapA prevents nonspecific DNA binding of RNAP without affecting the binding of promoter DNA recognition σ factor, thereby enhancing RNAP recycling.
- Department of Biochemistry and Molecular Biology, The Center for RNA Molecular Biology, The Center for Structural Biology, Pennsylvania State University, University Park, Pennsylvania, USA.
Organizational Affiliation: