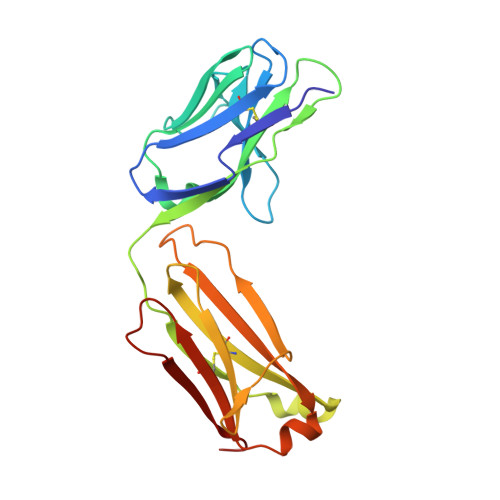
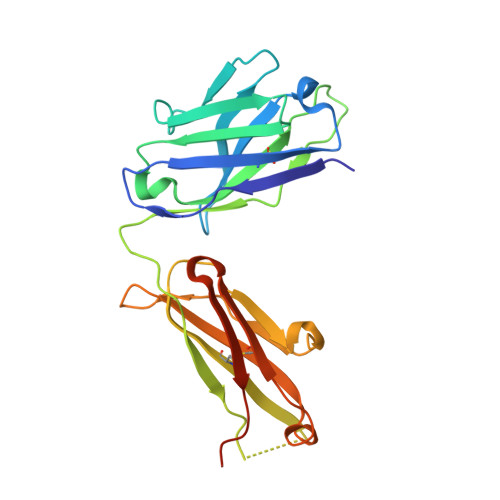
Structural basis of synergistic neutralization of Crimean-Congo hemorrhagic fever virus by human antibodies.
Mishra, A.K., Hellert, J., Freitas, N., Guardado-Calvo, P., Haouz, A., Fels, J.M., Maurer, D.P., Abelson, D.M., Bornholdt, Z.A., Walker, L.M., Chandran, K., Cosset, F.L., McLellan, J.S., Rey, F.A.(2022) Science 375: 104-109
- PubMed: 34793197 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.abl6502
- Primary Citation Related Structures:
7A59, 7A5A, 7KX4, 7L7R - PubMed Abstract:
Crimean-Congo hemorrhagic fever virus (CCHFV) is the most widespread tick-borne zoonotic virus, with a 30% case fatality rate in humans. Structural information is lacking in regard to the CCHFV membrane fusion glycoprotein Gc—the main target of the host neutralizing antibody response—as well as antibody–mediated neutralization mechanisms. We describe the structure of prefusion Gc bound to the antigen-binding fragments (Fabs) of two neutralizing antibodies that display synergy when combined, as well as the structure of trimeric, postfusion Gc. The structures show the two Fabs acting in concert to block membrane fusion, with one targeting the fusion loops and the other blocking Gc trimer formation. The structures also revealed the neutralization mechanism of previously reported antibodies against CCHFV, providing the molecular underpinnings essential for developing CCHFV–specific medical countermeasures for epidemic preparedness.
- Department of Molecular Biosciences, The University of Texas at Austin, Austin, TX 78712, USA.
Organizational Affiliation: