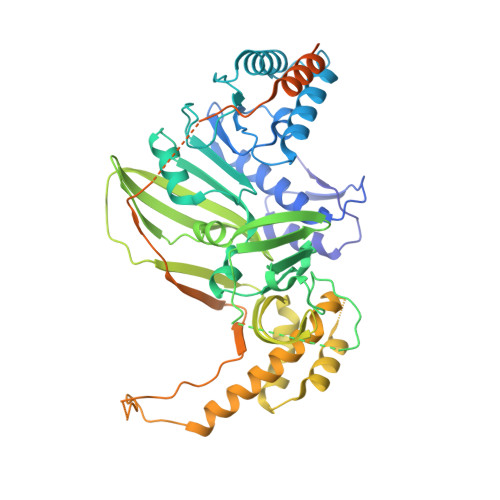
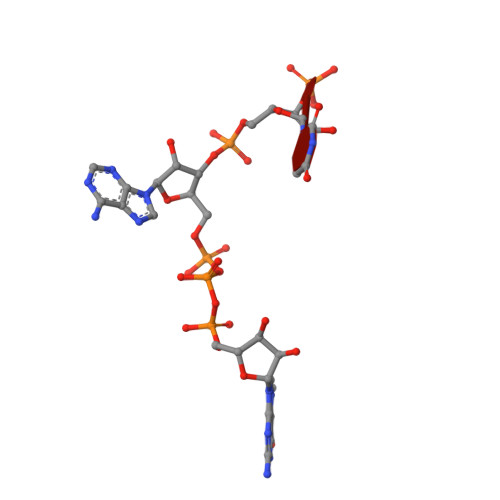
Molecular basis of specific viral RNA recognition and 5'-end capping by the Chikungunya virus nsP1.
Zhang, K., Law, M.C.Y., Nguyen, T.M., Tan, Y.B., Wirawan, M., Law, Y.S., Jeong, L.S., Luo, D.(2022) Cell Rep 40: 111133-111133
- PubMed: 35905713 Search on PubMed
- DOI: https://doi.org/10.1016/j.celrep.2022.111133
- Primary Citation Related Structures:
7FGG, 7FGH, 7FGI, 7X01 - PubMed Abstract:
Many viruses encode RNA-modifying enzymes to edit the 5' end of viral RNA to mimic the cellular mRNA for effective protein translation, genome replication, and evasion of the host defense mechanisms. Alphavirus nsP1 synthesizes the 5' end Cap-0 structure of viral RNAs. However, the molecular basis of the capping process remains unclear. We determine high-resolution cryoelectron microscopy (cryo-EM) structures of Chikungunya virus nsP1 in complex with m7GTP/SAH, covalently attached m7GMP, and Cap-0 viral RNA. These structures reveal details of viral-RNA-capping reactions and uncover a sequence-specific virus RNA-recognition pattern that, in turn, regulates viral-RNA-capping efficiency to ensure optimal genome replication and subgenomic RNA transcription. This sequence-specific enzyme-RNA pairing is conserved across all alphaviruses.
- Lee Kong Chian School of Medicine, Nanyang Technological University, EMB 03-07, 59 Nanyang Drive, Singapore 636921, Singapore; NTU Institute of Structural Biology, Nanyang Technological University, EMB 06-01, 59 Nanyang Drive, Singapore 636921, Singapore; School of Medicine, Southern University of Science and Technology, Shenzhen, Guangdong 518055, China.
Organizational Affiliation: