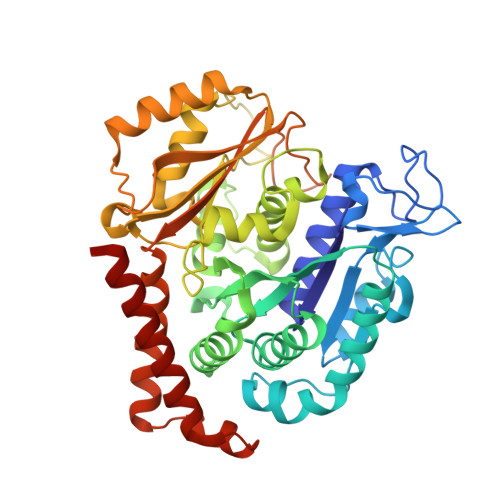
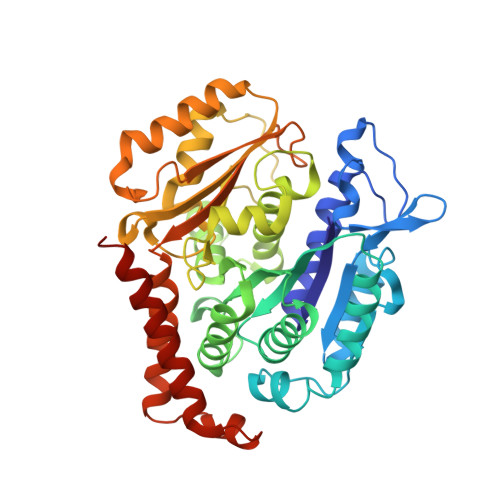
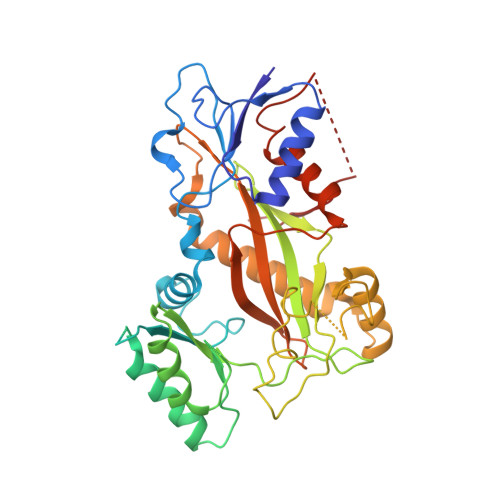
C3 ester side chain plays a pivotal role in the antitumor activity of Maytansinoids.
Li, W., Huang, M., Li, Y., Xia, A., Tan, L., Zhang, Z., Wang, Y., Yang, J.(2021) Biochem Biophys Res Commun 566: 197-203
- PubMed: 34144258 Search on PubMed
- DOI: https://doi.org/10.1016/j.bbrc.2021.05.071
- Primary Citation Related Structures:
7E4Q, 7E4R, 7E4Z - PubMed Abstract:
Maytansinoids, the chemical derivatives of Maytansine, are commonly used as potent cytotoxic payloads in antibody-drug conjugates (ADC). Structure-activity-relationship studies had identified the C3 ester side chain as a critical element for antitumor activity of maytansinoids. The maytansinoids bearing the methyl group at C3 position with D configuration were about 100 to 400-fold less cytotoxic than their corresponding L-epimers toward various cell lines. The detailed mechanism of how chirality affects the anticancer activity remains elusive. In this study, we determined the high-resolution crystal structure of tubulin in complex with maytansinol, L-DM1-SMe and D-DM1-SMe. And we found the carbonyl oxygen atom of the ester moiety and the tail thiomethyl group at C3 side chain of L-DM1-SMe form strong intramolecular interaction with the hydroxyl at position 9 and the benzene ring, respectively, fixing the bioactive conformation and enhancing the binding affinity. Additionally, ligand-based and structure-based virtually screening methods were used to screen the commercially macrocyclic compounds library, and 15 macrocyclic structures were picketed out as putatively new maytansine-site inhibitors. Our study provides a possible strategy for the rational discovery of next-generation maytansine site inhibitors.
- State Key Laboratory of Biotherapy and Collaborative Innovation Center for Biotherapy, West China Hospital, Sichuan University, Chengdu, 610041, PR China. Electronic address: liwentingo@qq.com.
Organizational Affiliation: