A Novel Family of Winged-Helix Single-Stranded DNA-Binding Proteins from Archaea.
Huang, C., Liu, X., Chen, Y., Zhou, J., Li, W., Ding, N., Huang, L., Chen, J., Zhang, Z.(2022) Int J Mol Sci 23
- PubMed: 35408816 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.3390/ijms23073455
- Primary Citation Related Structures:
7BZH - PubMed Abstract:
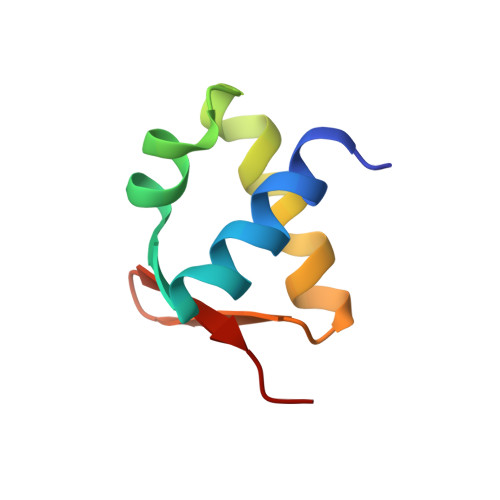
The winged helix superfamily comprises a large number of structurally related nucleic acid-binding proteins. While these proteins are often shown to bind dsDNA, few are known to bind ssDNA. Here, we report the identification and characterization of Sul7s, a novel winged-helix single-stranded DNA binding protein family highly conserved in Sulfolobaceae . Sul7s from Sulfolobus islandicus binds ssDNA with an affinity approximately 15-fold higher than that for dsDNA in vitro. It prefers binding oligo(dT) 30 over oligo(dC) 30 or a dG-rich 30-nt oligonucleotide, and barely binds oligo(dA) 30 . Further, binding by Sul7s inhibits DNA strand annealing, but shows little effect on the melting temperature of DNA duplexes. The solution structure of Sul7s determined by NMR shows a winged helix-turn-helix fold, consisting of three α-helices, three β-strands, and two short wings. It interacts with ssDNA via a large positively charged binding surface, presumably resulting in ssDNA deformation. Our results shed significant light on not only non-OB fold single-stranded DNA binding proteins in Archaea, but also the divergence of the winged-helix proteins in both function and structure during evolution.
- MOE Key Laboratory of Precision Nutrition and Food Quality, College of Food Science and Nutritional Engineering, China Agricultural University, Beijing 100083, China.
Organizational Affiliation: