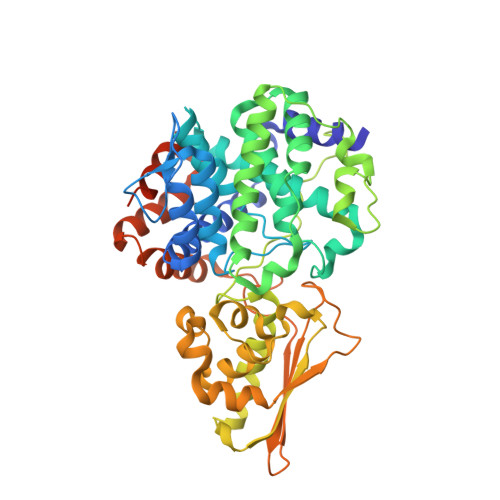
The crystal structure of mouse IRG1 suggests that cis-aconitate decarboxylase has an open and closed conformation.
Chun, H.L., Lee, S.Y., Kim, K.H., Lee, C.S., Oh, T.J., Park, H.H.(2020) PLoS One 15: e0242383-e0242383
- PubMed: 33259501 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1371/journal.pone.0242383
- Primary Citation Related Structures:
7BR9 - PubMed Abstract:
Coordinate and structural factors were deposited with the Protein Data Bank under PDB ID: 7BR9.
- College of Pharmacy, Chung-Ang University, Seoul, Republic of Korea.
Organizational Affiliation: