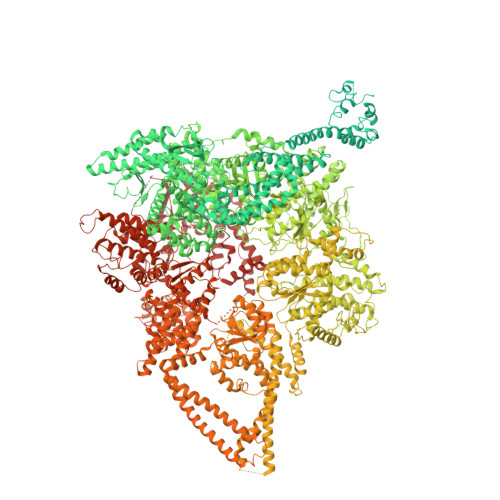
Shulin packages axonemal outer dynein arms for ciliary targeting.
Mali, G.R., Ali, F.A., Lau, C.K., Begum, F., Boulanger, J., Howe, J.D., Chen, Z.A., Rappsilber, J., Skehel, M., Carter, A.P.(2021) Science 371: 910-916
- PubMed: 33632841 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.abe0526
- Primary Citation Related Structures:
6ZYW, 6ZYX, 6ZYY - PubMed Abstract:
The main force generators in eukaryotic cilia and flagella are axonemal outer dynein arms (ODAs). During ciliogenesis, these ~1.8-megadalton complexes are assembled in the cytoplasm and targeted to cilia by an unknown mechanism. Here, we used the ciliate Tetrahymena to identify two factors (Q22YU3 and Q22MS1) that bind ODAs in the cytoplasm and are required for ODA delivery to cilia. Q22YU3, which we named Shulin, locked the ODA motor domains into a closed conformation and inhibited motor activity. Cryo-electron microscopy revealed how Shulin stabilized this compact form of ODAs by binding to the dynein tails. Our findings provide a molecular explanation for how newly assembled dyneins are packaged for delivery to the cilia.
- MRC Laboratory of Molecular Biology, Francis Crick Avenue, Cambridge CB2 0QH, UK.
Organizational Affiliation: