Fragment of human fibronectin containing the 11th type III domain, extra domain A, and 12th type III domain
Mou, T.C., Nepomuceno, P.A., Sprang, S.R., Briknarova, K.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Fibronectin | A [auth B], B [auth A] | 277 | Homo sapiens | Mutation(s): 0 Gene Names: FN1, FN | 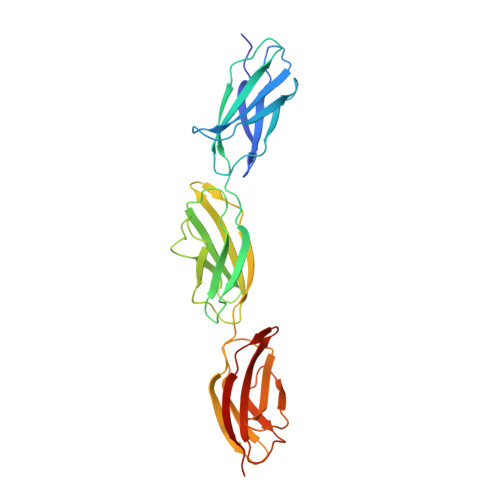 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P02751 GTEx: ENSG00000115414 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P02751 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| GOL Download:Ideal Coordinates CCD File | C [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 50.479 | α = 90 |
| b = 76.42 | β = 104.09 |
| c = 96.796 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | NIH P20 GM103546 |
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | NIH 5R01 GM114657 |