X-ray Structure of SARS-CoV-2 main protease bound to Boceprevir at 1.45 A
Anson, B., Mesecar, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 3C-like proteinase | 306 | Severe acute respiratory syndrome coronavirus 2 | Mutation(s): 0 Gene Names: rep, 1a-1b EC: 3.4.22.69 | 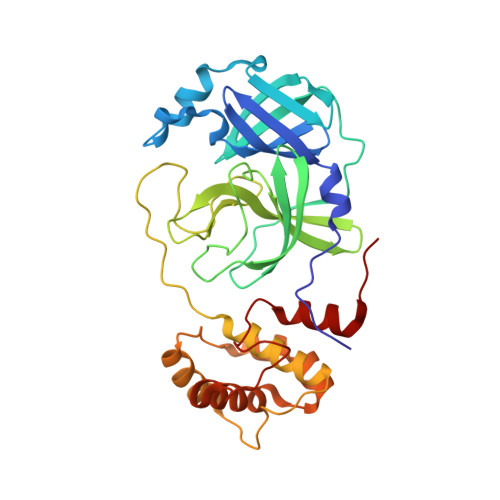 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0DTD1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| U5G Download:Ideal Coordinates CCD File | B [auth A] | boceprevir (bound form) C27 H47 N5 O5 FEBWCINGHXXUCV-GFLQDLJJSA-N |  | ||
| Entity ID: 2 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_002382 (U5G) Query on PRD_002382 | B [auth A] | Boceprevir (bound form) | Peptide-like / Enzyme inhibitor |  | |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 97.549 | α = 90 |
| b = 81.086 | β = 116.979 |
| c = 54.669 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| SCALEPACK | data scaling |
| PHENIX | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute Of Allergy and Infectious Diseases (NIH/NIAID) | United States | HHSN272201700060C |