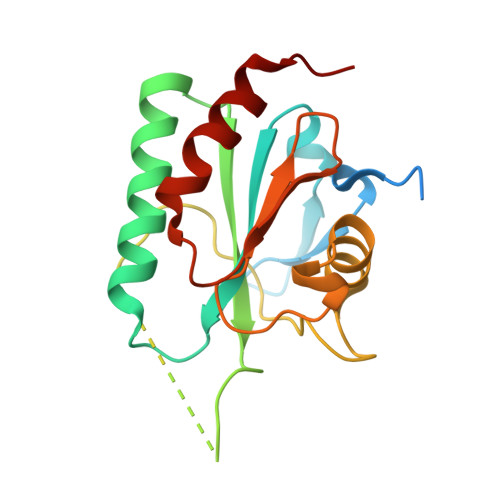
Structural and functional characterization of the glutathione peroxidase-like thioredoxin peroxidase from the fungus Trichoderma reesei.
Adriani, P.P., de Paiva, F.C.R., de Oliveira, G.S., Leite, A.C., Sanches, A.S., Lopes, A.R., Dias, M.V.B., Chambergo, F.S.(2020) Int J Biol Macromol 167: 93-100
- PubMed: 33259843 Search on PubMed
- DOI: https://doi.org/10.1016/j.ijbiomac.2020.11.179
- Primary Citation Related Structures:
6VPD - PubMed Abstract:
Glutathione peroxidases (GPx) are a family of enzymes with the ability to reduce organic and inorganic hydroperoxides to the corresponding alcohols using glutathione or thioredoxin as an electron donor. Here, we report the functional and structural characterization of a GPx identified in Trichoderma reesei (TrGPx). TrGPx was recombinantly expressed in a bacterial host and purified using affinity. Using a thioredoxin coupled assay, TrGPx exhibited activity of 28 U and 12.5 U in the presence of the substrates H 2 O 2 and t-BOOH, respectively, and no activity was observed when glutathione was used. These results indicated that TrGPx is a thioredoxin peroxidase and hydrolyses H 2 O 2 better than t-BOOH. TrGPx kinetic parameters using a pyrogallol assay resulted at K mapp = 11.7 mM , V maxapp = 10.9 IU/μg TrGPx, k cat = 19 s -1 and a catalytic efficiency of 1.6 mM -1 s -1 to H 2 O 2 as substrate. Besides that, TrGPx demonstrated an optimum pH ranging from 9.0-12.0 and a half-life of 36 min at 80 °C. TrGPx 3D-structure was obtained in a reduced state and non-catalytic conformation. The overall fold is similar to the other phospholipid-hydroperoxide glutathione peroxidases. These data contribute to understand the antioxidant mechanism in fungi and provide information for using antioxidant enzymes in biotechnological applications.
- Escola de Artes, Ciências e Humanidades, Universidade de São Paulo, 1000 Arlindo Bettio Avenue, São Paulo, Brazil.
Organizational Affiliation: