Protein-Induced Change in Ligand Protonation during Trypsin and Thrombin Binding: Hint on Differences in Selectivity Determinants of Both Proteins?
Ngo, K., Collins-Kautz, C., Gerstenecker, S., Wagner, B., Heine, A., Klebe, G.(2020) J Med Chem 63: 3274-3289
- PubMed: 32011145 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jmedchem.9b02061
- Primary Citation Related Structures:
6HSX, 6SY3, 6T0M, 6T0P, 6T3Q, 6T4A, 6T5W, 6TDT - PubMed Abstract:
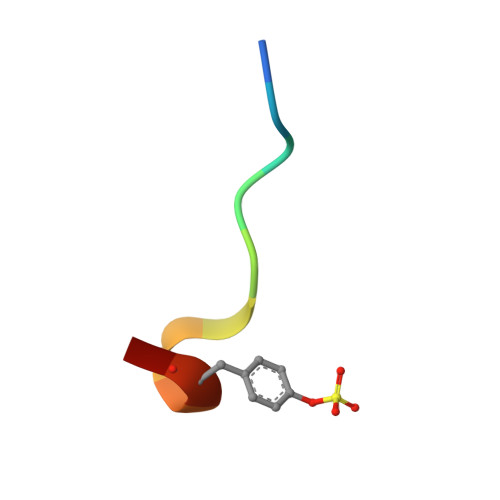
Trypsin and thrombin, structurally similar serine proteases, recognize different substrates; thrombin cleaves after Arg, whereas trypsin cleaves after Lys/Arg. Both recognize basic substrate headgroups via Asp189 at the bottom of the S1 pocket. By crystallography and isothermal titration calorimetry (ITC), we studied a series of d-Phe/d-DiPhe-Pro-(amino)pyridines. Identical ligand pairs show the same binding poses. Surprisingly, one ligand binds to trypsin in protonated state and to thrombin in unprotonated state at P1 along with differences in the residual solvation pattern. While trypsin binding is mediated by an ordered water molecule, in thrombin, water is scattered over three hydration sites. Although having highly similar S1 pockets, our results suggest different electrostatic properties of Asp189 possibly contributing to the selectivity determinant. Thrombin binds a specific Na + ion next to Asp189, which is absent in trypsin. The electrostatic properties across the S1 pocket are further attenuated by charged Glu192 at the rim of S1 in thrombin, which is replaced by uncharged Gln192 in trypsin.
- Institute of Pharmaceutical Chemistry, Philipps-University Marburg, Marbacher Weg 6, 35032 Marburg, Germany.
Organizational Affiliation: