Biomacromolecular charge chirality detected using chiral plasmonic nanostructures
Rodier, M., Keijzer, C., Milner, J., Karimullah, A., Roszak, A.W., Barron, L.D., Gadegaard, N., Lapthorn, A.J., Kadodwala, M.(2020) Nanoscale Horiz
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2020) Nanoscale Horiz
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| 3-dehydroquinate dehydratase | 146 | Cutibacterium acnes | Mutation(s): 0 Gene Names: aroQ, CHT92_10435, CP876_09935 EC: 4.2.1.10 | 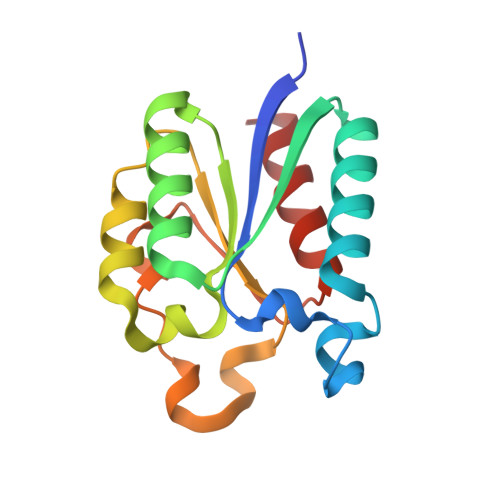 | |
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | E [auth A] F [auth A] H [auth B] J [auth C] K [auth C] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| GOL Download:Ideal Coordinates CCD File | G [auth A], I [auth B], M [auth C] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 128.82 | α = 90 |
| b = 128.82 | β = 90 |
| c = 75.999 | γ = 120 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| XDS | data reduction |
| Aimless | data scaling |
| PHASER | phasing |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Engineering and Physical Sciences Research Council | United Kingdom | EP/P00086X/1 |