Deciphering de molecular and structural interaction between IRF3 and HPV16 E6
Poirson, J., Suarez, I.P., Cousido-Siah, A., Forster, A., Chebaro, Y., Mitschler, A., Straub, M., Altschuh, D., Podjarny, A., Trave, G., Masson, M.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Maltose/maltodextrin-binding periplasmic protein,Interferon regulatory factor 3 | 383 | Escherichia coli, Homo sapiens This entity is chimeric | Mutation(s): 3 Gene Names: malE, b4034, JW3994, IRF3 | 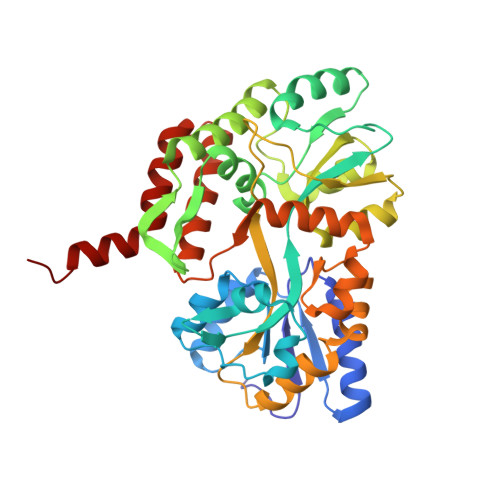 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q14653 GTEx: ENSG00000126456 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Groups | Q14653P0AEX9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein E6 | 153 | Human papillomavirus 16 | Mutation(s): 5 Gene Names: E6 | 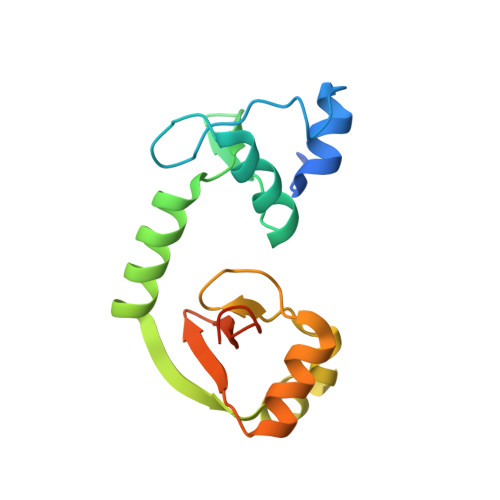 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P03126 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZN (Subject of Investigation/LOI) Download:Ideal Coordinates CCD File | D [auth B], E [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 97.659 | α = 90 |
| b = 132.87 | β = 90 |
| c = 43.025 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| French League Against Cancer | France | equipe labellisee 2015 and fellowship AB |
| National Institutes of Health/National Cancer Institute (NIH/NCI) | United States | R01CA134737 |
| Foundation for Medical Research (France) | France | fellowship to IPS |