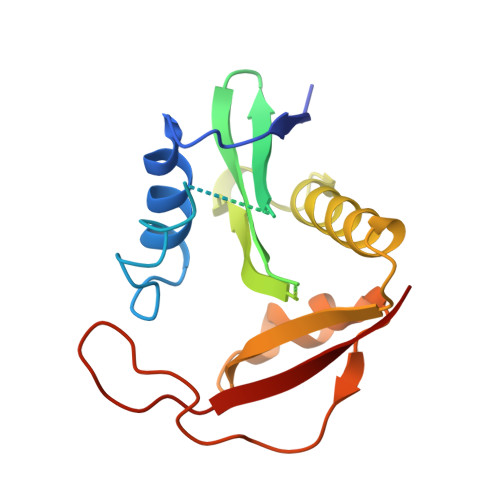
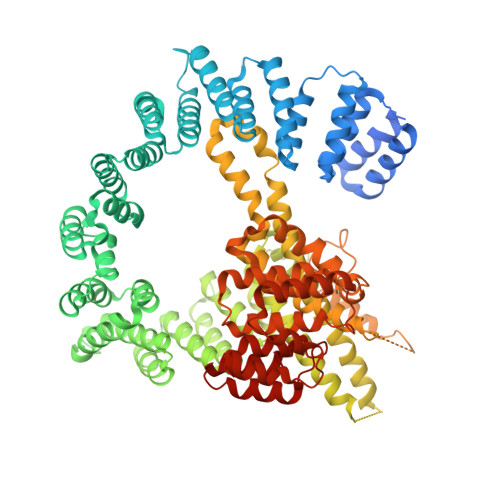
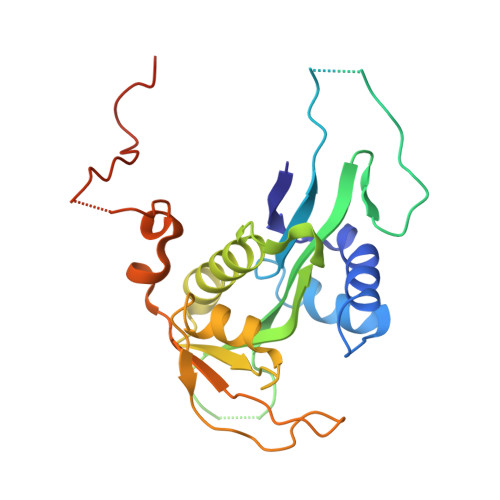
Structure and Mechanism of Acetylation by the N-Terminal Dual Enzyme NatA/Naa50 Complex.
Deng, S., Magin, R.S., Wei, X., Pan, B., Petersson, E.J., Marmorstein, R.(2019) Structure 27: 1057
- PubMed: 31155310 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2019.04.014
- Primary Citation Related Structures:
6O07 - PubMed Abstract:
NatA co-translationally acetylates the N termini of over 40% of eukaryotic proteins and can associate with another catalytic subunit, Naa50, to form a ternary NatA/Naa50 dual enzyme complex (also called NatE). The molecular basis of association between Naa50 and NatA and the mechanism for how their association affects their catalytic activities in yeast and human are poorly understood. Here, we determined the X-ray crystal structure of yeast NatA/Naa50 as a scaffold to understand coregulation of NatA/Naa50 activity in both yeast and human. We find that Naa50 makes evolutionarily conserved contacts to both the Naa10 and Naa15 subunits of NatA. These interactions promote catalytic crosstalk within the human complex, but do so to a lesser extent in the yeast complex, where Naa50 activity is compromised. These studies have implications for understanding the role of the NatA/Naa50 complex in modulating the majority of the N-terminal acetylome in diverse species.
- Department of Chemistry, University of Pennsylvania, 231 South 34(th) Street, Philadelphia, PA 19104, USA; Abramson Family Cancer Research Institute, Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA 19104, USA.
Organizational Affiliation: