X-ray crystallographic studies on the hydrogen isotope effects of green fluorescent protein at sub-angstrom resolutions
Tai, Y., Takaba, K., Hanazono, Y., Dao, H.A., Miki, K., Takeda, K.(2019) Acta Crystallogr D Biol Crystallogr 75: 1096-1106
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
(2019) Acta Crystallogr D Biol Crystallogr 75: 1096-1106
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Green fluorescent protein | 229 | Aequorea victoria | Mutation(s): 4 Gene Names: GFP | 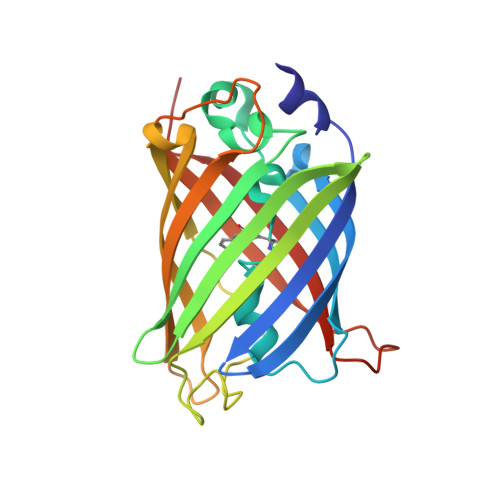 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P42212 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CRO Query on CRO | A | L-PEPTIDE LINKING | C15 H17 N3 O5 |  | THR, TYR, GLY |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 50.915 | α = 90 |
| b = 62.107 | β = 90 |
| c = 68.865 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| SHELXL | refinement |
| DENZO | data reduction |
| MOLREP | phasing |
| PDB_EXTRACT | data extraction |
| SCALEPACK | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Japan Society for the Promotion of Science | Japan | 17H03643 |
| Ministry of Education, Culture, Sports, Science and Technology (Japan) | Japan | Photon and Quantum Basic Research Coordinated Development Program |