scFv AbVance: increasing our knowledge of antibody structural space to enable faster and better decision making in drug discovery
Hargreaves, D.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| scFv AbVance: increasing our knowledge of antibody structural space to enable faster and better decision making in drug discovery | A [auth B] | 251 | Homo sapiens | Mutation(s): 0 | 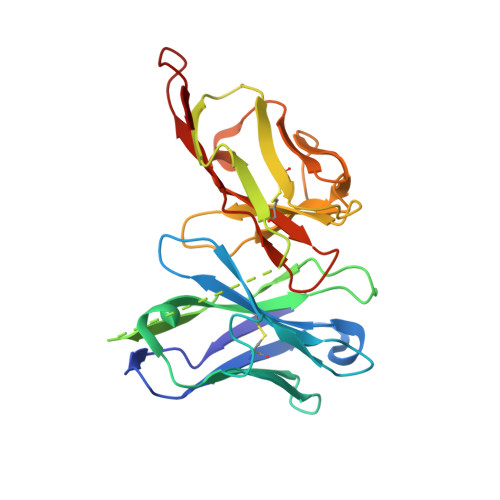 |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 45.396 | α = 90 |
| b = 70.033 | β = 90 |
| c = 78.468 | γ = 90 |
| Software Name | Purpose |
|---|---|
| BUSTER | refinement |
| XDS | data reduction |
| XDS | data scaling |
| PHASER | phasing |