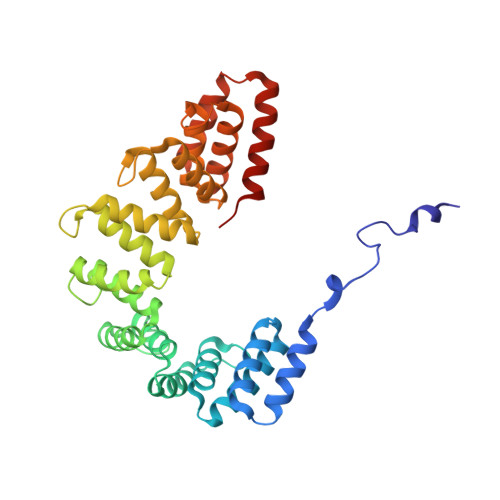
The structure of Legionella effector protein LpnE provides insights into its interaction with Oculocerebrorenal syndrome of Lowe (OCRL) protein.
Voth, K.A., Chung, I.Y.W., van Straaten, K., Li, L., Boniecki, M.T., Cygler, M.(2019) FEBS J 286: 710-725
- PubMed: 30479037
- DOI: https://doi.org/10.1111/febs.14710
- Primary Citation of Related Structures:
6DEH - PubMed Abstract:
Legionella pneumophila is a freshwater bacterium that replicates in predatory amoeba and alveolar macrophage. The ability of L. pneumophila to thrive in eukaryotic host cells is conferred by the Legionella containing vacuole (LCV). Formation and intracellular trafficking of the LCV are governed by an arsenal of effector proteins, many of which are secreted by the Icm/Dot Type 4 Secretion System. One such effector, known as LpnE (L. pneumophila Entry), has been implicated in facilitating bacterial entry into host cells, LCV trafficking, and substrate translocation. LpnE belongs to a subfamily of tetratricopeptide repeat proteins known as Sel1-like repeats (SLRs). All eight of the predicted SLRs in LpnE are required to promote host cell invasion. Herein, we report that LpnE(1-375) localizes to cis-Golgi in HEK293 cells via its signal peptide (aa 1-22). We further verify the interaction of LpnE(73-375) and LpnE(22-375) with Oculocerebrorenal syndrome of Lowe protein (OCRL) residues 10-208, restricting the known interacting residues for both proteins. To further characterize the SLR region of LpnE, we solved the crystal structure of LpnE(73-375) to 1.75Å resolution. This construct comprises all SLRs, which are arranged in a superhelical fold. The α-helices forming the inner concave surface of the LpnE superhelix suggest a potential protein-protein interaction interface. DATABASE: Coordinates and structure factors were deposited in the Protein Data Bank with the accession number 6DEH.
Organizational Affiliation:
Department of Biochemistry, University of Saskatchewan, Saskatoon, Canada.