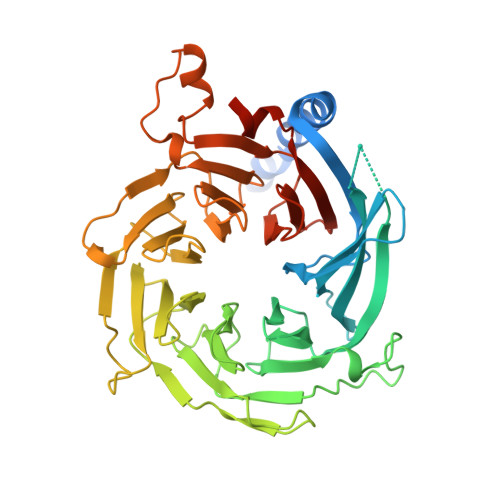
Structural and biochemical insights into human zinc finger protein AEBP2 reveals interactions with RBBP4
Sun, A., Li, F., Liu, Z., Jiang, Y., Zhang, J., Wu, J., Shi, Y.(2017) Protein Cell
- PubMed: 29134516 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s13238-017-0483-6
- Primary Citation Related Structures:
5Y0U, 5Y1U - Hefei National Laboratory for Physical Sciences at Microscale and School of Life Sciences, University of Science and Technology of China, Hefei, 230027, China.
Organizational Affiliation: