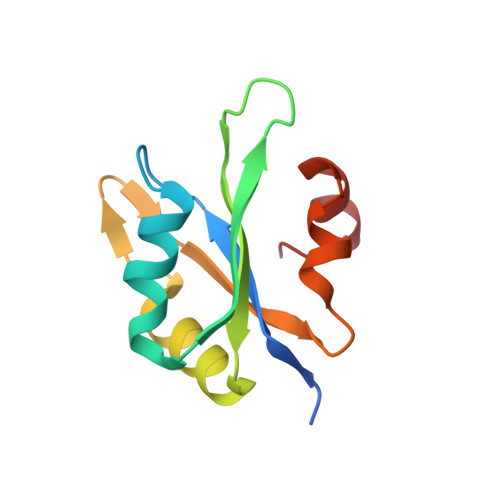
Structural analysis reveals the flexible C-terminus of Nop15 undergoes rearrangement to recognize a pre-ribosomal RNA folding intermediate.
Zhang, J., Gonzalez, L.E., Hall, T.M.T.(2017) Nucleic Acids Res 45: 2829-2837
- PubMed: 27789691 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkw961
- Primary Citation Related Structures:
5T9P - PubMed Abstract:
The RNA recognition motif (RRM) is the most abundant RNA-binding domain in eukaryotes, and it plays versatile roles in RNA metabolism. Despite its abundance, diversity of RRM structure and function is generated by variations on a conserved core. Yeast Nop15 is an RRM protein that is essential for large ribosomal subunit biogenesis. We determined a 2.0 Å crystal structure of Nop15 that reveals a C-terminal α-helical region obscures its canonical RNA-binding surface. Small-angle X-ray scattering, NMR and RNA-binding analyses further reveal that the C-terminal residues of Nop15 are highly flexible, but essential for tight RNA binding. Moreover, comparison with a recently reported cryo-electron microscopy structure indicates that dramatic rearrangement of the C-terminal region of Nop15 in the pre-ribosome exposes the RNA-binding surface to recognize the base of its stem-loop target RNA and extends a newly-formed α helix to the distal loop where it forms protein interactions.
- Epigenetics and Stem Cell Biology Laboratory, National Institute of Environmental Health Sciences, National Institutes of Health, P.O. Box 12233, MD F3-05, Research Triangle Park, NC 27709, USA.
Organizational Affiliation: