Redox-switchable siderophore anchor enables reversible artificial metalloenzyme assembly
Raines, A.D.J., Clarke, J.E., Blagova, E., Dodson, E.J., Wilson, K.S., Duhme-Klair, A.K.(2018) Nat Catal
Experimental Data Snapshot
Starting Model: experimental
View more details
(2018) Nat Catal
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Enterochelin ABC transporter substrate-binding protein | 288 | Campylobacter jejuni | Mutation(s): 0 Gene Names: AD53_07815, BKM79_06765 | 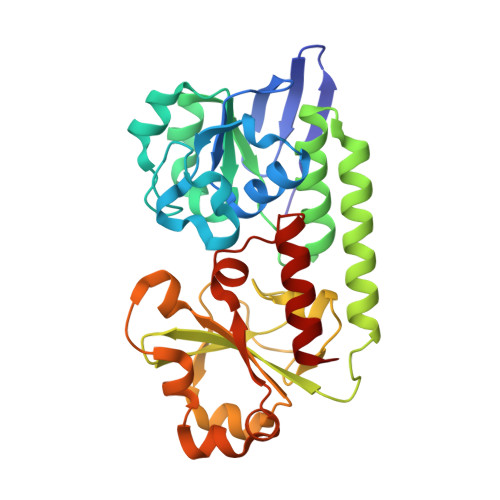 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q0P8Q4 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 95B Download:Ideal Coordinates CCD File | E [auth A], G [auth B] | Azotochelin C20 H22 N2 O8 KQPFLOCEYZIIRD-ZDUSSCGKSA-N |  | ||
| FE Download:Ideal Coordinates CCD File | D [auth A], F [auth B] | FE (III) ION Fe VTLYFUHAOXGGBS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 58.139 | α = 82.71 |
| b = 63.41 | β = 76.36 |
| c = 67.674 | γ = 79.03 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| Aimless | data scaling |
| MOLREP | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Engineering and Physical Sciences Research Council | United Kingdom | -- |