Arsenite oxidase AioAB from Rhizobium sp. str. NT-26 mutant AioBF108A
Santos-Silva, T., Romao, M., Vieira, M., Marques, A.T.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| AroA | 843 | Pseudorhizobium banfieldiae | Mutation(s): 0 Gene Names: aroA, aioA, NT26_p10030 EC: 1.20.98.1 | 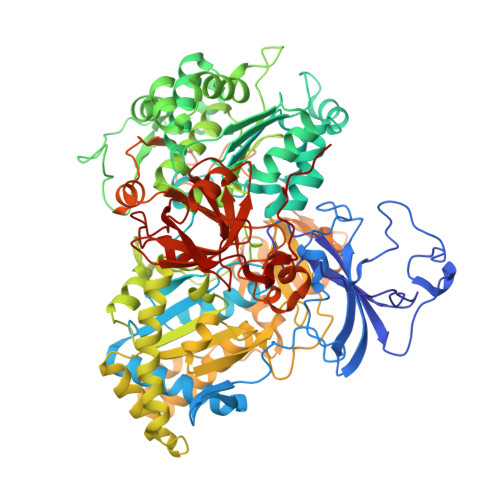 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q6VAL8 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Arsenite oxidase small subunit AioB Rieske [2Fe-2S] cluster | 132 | Pseudorhizobium banfieldiae | Mutation(s): 0 Gene Names: aioB, NT26_p10029 EC: 1.20.98.1 | 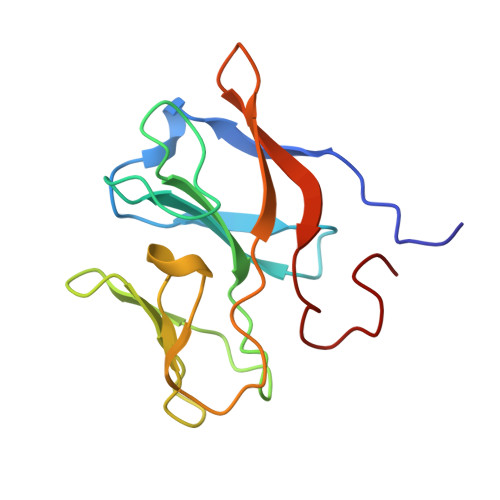 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | L0NMC5 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 11 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| MGD Download:Ideal Coordinates CCD File | I [auth A] IA [auth E] J [auth A] JA [auth E] RA [auth G] | 2-AMINO-5,6-DIMERCAPTO-7-METHYL-3,7,8A,9-TETRAHYDRO-8-OXA-1,3,9,10-TETRAAZA-ANTHRACEN-4-ONE GUANOSINE DINUCLEOTIDE C20 H26 N10 O13 P2 S2 VQAGYJCYOLHZDH-ILXWUORBSA-N |  | ||
| P33 Download:Ideal Coordinates CCD File | YA [auth G], ZA [auth G] | 3,6,9,12,15,18-HEXAOXAICOSANE-1,20-DIOL C14 H30 O8 XPJRQAIZZQMSCM-UHFFFAOYSA-N |  | ||
| F3S Download:Ideal Coordinates CCD File | M [auth A], MA [auth E], VA [auth G], Z [auth C] | FE3-S4 CLUSTER Fe3 S4 FCXHZBQOKRZXKS-UHFFFAOYSA-N |  | ||
| FES Download:Ideal Coordinates CCD File | DB [auth H], HA [auth D], QA [auth F], T [auth B] | FE2/S2 (INORGANIC) CLUSTER Fe2 S2 NIXDOXVAJZFRNF-UHFFFAOYSA-N |  | ||
| PGE Download:Ideal Coordinates CCD File | R [auth A] | TRIETHYLENE GLYCOL C6 H14 O4 ZIBGPFATKBEMQZ-UHFFFAOYSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | EA [auth C] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | AA [auth C] BA [auth C] CA [auth C] DA [auth C] N [auth A] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| 4MO Download:Ideal Coordinates CCD File | L [auth A], LA [auth E], UA [auth G], Y [auth C] | MOLYBDENUM(IV) ION Mo ZIKKVZAYJJZBGE-UHFFFAOYSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | AB [auth G], BB [auth G], FA [auth C], PA [auth E], S [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | CB [auth G], GA [auth C], U [auth B] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| O Download:Ideal Coordinates CCD File | K [auth A], KA [auth E], TA [auth G], X [auth C] | OXYGEN ATOM O XLYOFNOQVPJJNP-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 141.408 | α = 90 |
| b = 148.665 | β = 90 |
| c = 232.009 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SCALEPACK | data scaling |
| REFMAC | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| REFMAC | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| FCT | Portugal | PTDC/BBB-BEP/1185/2014 |
| FCT | Portugal | UID/Multi/04378/2013 |
| FCT | Portugal | PTDC/QEQ-MED/1902/2014 |
| PT2020 | Portugal | POCI-01-0145-FEDER-007728 |