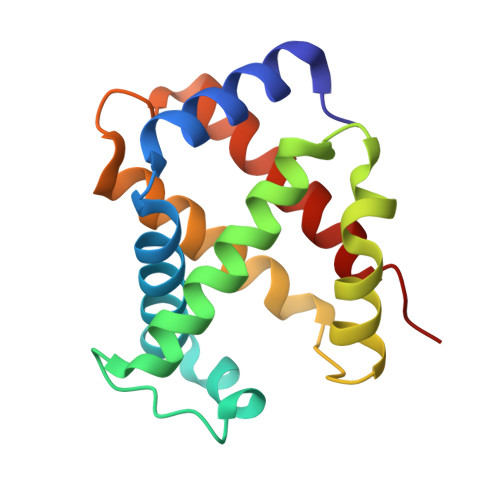
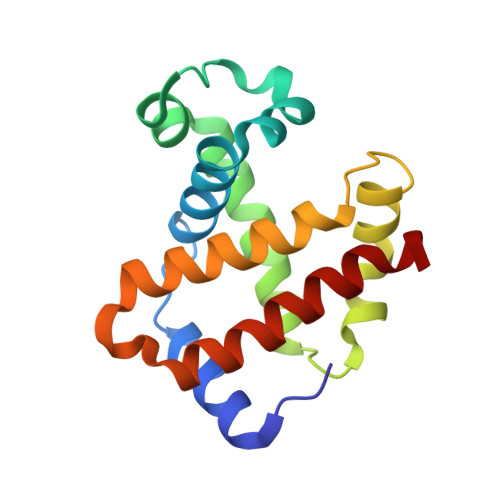
Crystal structure of carbonmonoxy sickle hemoglobin in R-state conformation.
Ghatge, M.S., Ahmed, M.H., Omar, A.S., Pagare, P.P., Rosef, S., Kellogg, G.E., Abdulmalik, O., Safo, M.K.(2016) J Struct Biol 194: 446-450
- PubMed: 27085422 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.jsb.2016.04.003
- Primary Citation Related Structures:
5E6E - PubMed Abstract:
The fundamental pathophysiology of sickle cell disease is predicated by the polymerization of deoxygenated (T-state) sickle hemoglobin (Hb S) into fibers that distort red blood cells into the characteristic sickle shape. The crystal structure of deoxygenated Hb S (DeoxyHb S) and other studies suggest that the polymer is initiated by a primary interaction between the mutation βVal6 from one Hb S molecule, and a hydrophobic acceptor pocket formed by the residues βAla70, βPhe85 and βLeu88 of an adjacent located Hb S molecule. On the contrary, oxygenated or liganded Hb S does not polymerize or incorporate in the polymer. In this paper we present the crystal structure of carbonmonoxy-ligated sickle Hb (COHb S) in the quaternary classical R-state at 1.76Å. The overall structure and the pathological donor and acceptor environments of COHb S are similar to those of the isomorphous CO-ligated R-state normal Hb (COHb A), but differ significantly from DeoxyHb S as expected. More importantly, the packing of COHb S molecules does not show the typical pathological interaction between βVal6 and the βAla70, βPhe85 and βLeu88 hydrophobic acceptor pocket observed in DeoxyHb S crystal. The structural analysis of COHb S, COHb A and DeoxyHb S provides atomic level insight into why liganded hemoglobin does not form a polymer.
- Department of Medicinal Chemistry, and The Institute for Structural Biology, Drug Discovery and Development, School of Pharmacy, Virginia Commonwealth University, Richmond, VA 23298, United States.
Organizational Affiliation: