Crystal structure of thioredoxin reductase from Cryptococcus neoformans in complex with FAD (FO conformation)
Bravo-Chaucanes, C.P., Valadares, N.F., Felipe, M.S.S., Barbosa, J.A.R.G.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Thioredoxin reductase | 371 | Cryptococcus neoformans H99 | Mutation(s): 0 Gene Names: CNAG_05847 EC: 1.8.1.9 | 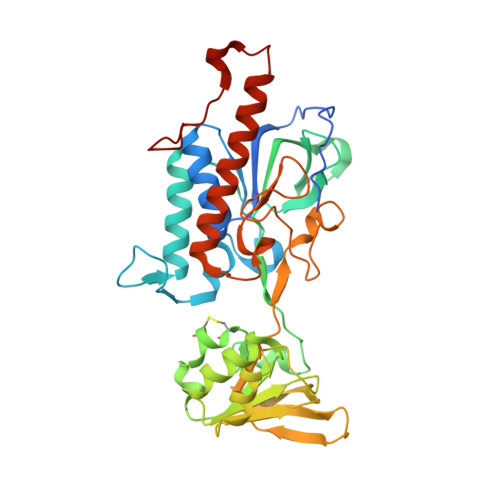 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | J9VRX9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD Download:Ideal Coordinates CCD File | C [auth A], H [auth B] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| PEG Download:Ideal Coordinates CCD File | E [auth A] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | D [auth A], I [auth B] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | G [auth A] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| CA Download:Ideal Coordinates CCD File | F [auth A], J [auth B], K [auth B] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 86.14 | α = 90 |
| b = 108.828 | β = 90 |
| c = 70.354 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| DENZO | data reduction |
| SCALEPACK | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Brazilian National Council for Scientific and Technological Development (CNPq) | Brazil | 564007/2010-2 |
| FAPDF/CNPq | Brazil | 193000569/2009 |