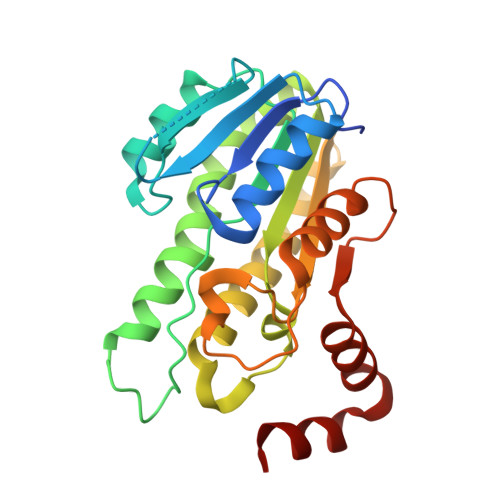
Crystal structure of a short-chain dehydrogenase from Burkholderia cenocepacia J2315 in complex with NADP+ and benzoic acid
Belfon, K.K.J., Beyer, O., Abendroth, J., Dranow, D.M., Lorimer, D.D., Abramov, A., Latimore, Y., Hamilton, C., Dawkins, A., Hinojosa, I., Martinez, X., Mirabel, S., Duncan, M., Womack, R., Hicks, L., Turlington, Z.R., Edwards, T.E., Torelli, A.T., Hicks, K.A., French, J.B.(2024) Acta Crystallogr F Struct Biol Commun 80