Crystal structure of Amidase, hydantoinase/carbamoylase family from Burkholderia multivorans
Abendroth, J., Delker, S.L., Lorimer, D.D., Edwards, T.E.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Deacylase | 434 | Burkholderia multivorans ATCC 17616 | Mutation(s): 0 Gene Names: BMULJ_06149 EC: 3.5.1.87 | 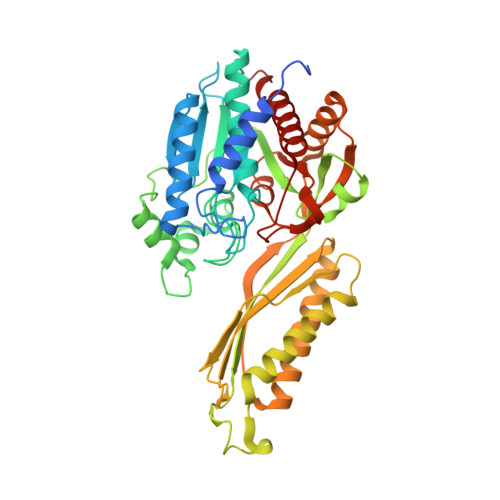 | |
UniProt | |||||
Find proteins for A0A0H3KRF1 (Burkholderia multivorans (strain ATCC 17616 / 249)) Explore A0A0H3KRF1 Go to UniProtKB: A0A0H3KRF1 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0H3KRF1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| URP Download:Ideal Coordinates CCD File | G [auth A], L [auth C] | N-(AMINOCARBONYL)-BETA-ALANINE C4 H8 N2 O3 JSJWCHRYRHKBBW-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | E [auth A] F [auth A] H [auth B] J [auth C] K [auth C] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| EDO Download:Ideal Coordinates CCD File | I [auth B], N [auth D] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 86.46 | α = 90 |
| b = 88.51 | β = 90.21 |
| c = 126.44 | γ = 90 |
| Software Name | Purpose |
|---|---|
| XDS | data scaling |
| XSCALE | data scaling |
| MOLREP | phasing |
| Coot | model building |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |