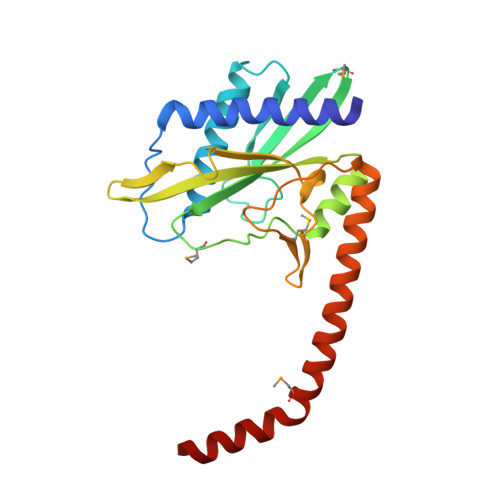
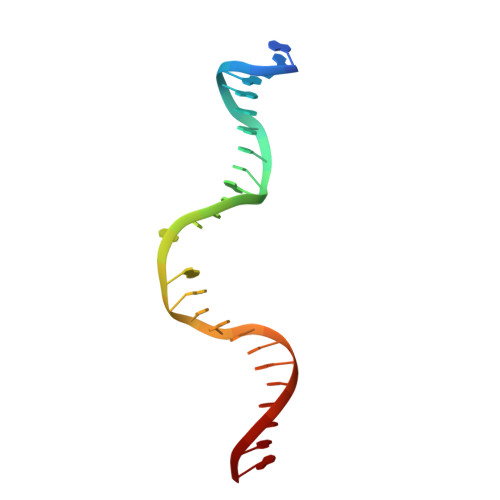
DNA recognition by the SwaI restriction endonuclease involves unusual distortion of an 8 base pair A:T-rich target.
Shen, B.W., Heiter, D.F., Lunnen, K.D., Wilson, G.G., Stoddard, B.L.(2017) Nucleic Acids Res 45: 1516-1528
- PubMed: 28180307 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkw1200
- Primary Citation Related Structures:
5TGQ, 5TGX, 5TH3 - PubMed Abstract:
R.SwaI, a Type IIP restriction endonuclease, recognizes a palindromic eight base pair (bp) symmetric sequence, 5΄-ATTTAAAT-3΄, and cleaves that target at its center to generate blunt-ended DNA fragments. Here, we report three crystal structures of SwaI: unbound enzyme, a DNA-bound complex with calcium ions; and a DNA-bound, fully cleaved complex with magnesium ions. We compare these structures to two structurally similar ‘PD-D/ExK’ restriction endonucleases (EcoRV and HincII) that also generate blunt-ended products, and to a structurally distinct enzyme (the HNH endonuclease PacI) that also recognizes an 8-bp target site consisting solely of A:T base pairs. Binding by SwaI induces an extreme bend in the target sequence accompanied by un-pairing and re-ordering of its central A:T base pairs. This result is reminiscent of a more dramatic target deformation previously described for PacI, implying that long A:T-rich target sites might display structural or dynamic behaviors that play a significant role in endonuclease recognition and cleavage.
- Division of Basic Sciences, Fred Hutchinson Cancer Research Center, 1100 Fairview Ave. North, Seattle, WA, USA.
Organizational Affiliation: